Antineutrophil cytoplasmic autoantibodies interact with primary granule constituents on the surface of apoptotic neutrophils in the absence of neutrophil priming
- PMID: 8976178
- PMCID: PMC2196384
- DOI: 10.1084/jem.184.6.2231
Antineutrophil cytoplasmic autoantibodies interact with primary granule constituents on the surface of apoptotic neutrophils in the absence of neutrophil priming
Abstract
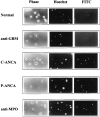
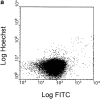
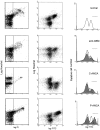
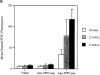
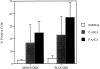
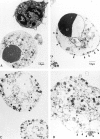
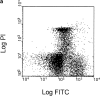
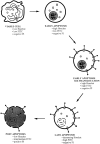
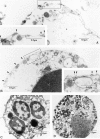
The pathogenic role of antineutrophil cytoplasmic autoantibodies (ANCA) remains controversial because of the difficulty in explaining how extracellular ANCA can interact with intracellular primary granule constituents. It has been postulated that cytokine priming of neutrophils (PMN), as may occur during a prodromal infection, is an important trigger for mobilization of granules to the cell surface, where they may interact with ANCA. We show by electron microscopy that apoptosis of unprimed PMN is also associated with the translocation of cytoplasmic granules to the cell surface and alignment just beneath an intact cell membrane. Immunofluorescent microscopy and FACS analysis demonstrate reactivity of ANCA-positive sera and antimyeloperoxidase antibodies with apoptotic PMN, but not with viable PMN. Moreover, we show that apoptotic PMN may be divided into two subsets, based on the presence or absence of granular translocation, and that surface immunogold labeling of myeloperoxidase occurs only in the subset of PMN showing translocation. These results provide a novel mechanism that is independent of priming, by which ANCA may gain access to PMN granule components during ANCA-associated vasculitis.
Figures






References
-
- Van der Woude, F.J., N. Rasmussen, S. Lobatto, A. Wiik, H. Permin, L.A. van Es, M. van der Giessen, G.K. van der Hem, and T.H. The. 1985. Autoantibodies against neutrophils and monocytes. Tools for diagnosis and marker of disease activity in Wegener's granulomatosis. Lancet. i:425–429. - PubMed
-
- Falk RJ, Jeannette JC. Anti-neutrophil cytoplasmic autoantibodies with specificity for myeloperoxidase in patients with systemic vasculitis and idiopathic necrotizing and crescentic glomerulonephritis. N Eng J Med. 1988;318:1651–1657. - PubMed
-
- Nölle B, Specks U, Lüdemann J, Rohrbach MS, DeRemee RA, Gross WL. Anticytoplasmic autoantibodies: their immunodiagnostic value in Wegener granulomatosis. Ann Int Med. 1989;111:28–40. - PubMed
-
- Cohen Tervaert, J.W., R. Goldschmeding, J.D. Elema, P.C. Limburg, M. van der Giessen, M.G. Huitema, M.I. Koolen, R.J. Hené, T.H. The, G.K. van der Hem, et al. Association of autoantibodies to myeloperoxidase with different forms of vasculitis. Arthritis Rheum. 1990;33:1264–1272. - PubMed
-
- Lesavre P. Antineutrophil cytoplasmic autoantibodies antigen specificity. Am J Kidney Dis. 1991;18:159–163. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

