Immune response in human melanoma after transfer of an allogeneic class I major histocompatibility complex gene with DNA-liposome complexes
- PMID: 8986821
- PMCID: PMC26414
- DOI: 10.1073/pnas.93.26.15388
Immune response in human melanoma after transfer of an allogeneic class I major histocompatibility complex gene with DNA-liposome complexes
Abstract
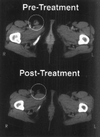
Analysis of the antitumor immune response after gene transfer of a foreign major histocompatibility complex class I protein, HLA-B7, was performed. Ten HLA-B7-negative patients with stage IV melanoma were treated in an effort to stimulate local tumor immunity. Plasmid DNA was detected within treated tumor nodules, and RNA encoding recombinant HLA-B7 or HLA-B7 protein was demonstrated in 9 of 10 patients. T cell migration into treated lesions was observed and tumor-infiltrating lymphocyte reactivity was enhanced in six of seven and two of two patients analyzed, respectively. In contrast, the frequency of cytotoxic T lymphocyte against autologous tumor in circulating peripheral blood lymphocytes was not altered significantly, suggesting that peripheral blood lymphocyte reactivity is not indicative of local tumor responsiveness. Local inhibition of tumor growth was detected after gene transfer in two patients, one of whom showed a partial remission. This patient subsequently received treatment with tumor-infiltrating lymphocytes derived from gene-modified tumor, with a complete regression of residual disease. Thus, gene transfer with DNA-liposome complexes encoding an allogeneic major histocompatibility complex protein stimulated local antitumor immune responses that facilitated the generation of effector cells for immunotherapy of cancer.
Figures


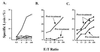
References
-
- Nabel G J, Nabel E G, Yang Z, Fox B A, Plautz G E, Gao X, Huang L, Shu S, Gordon D, Chang A E. Cold Spring Harbor Symp Quant Biol. 1994;49:699–707. - PubMed
-
- Felgner J H, Kumar R, Sridhar C N, Wheeler C J, Tsai Y J, Border R, Ramsey P, Martin M, Felgner P L. J Biol Chem. 1994;269:2550–2561. - PubMed
-
- San H, Yang Z, Pompili V J, Jaffe M L, Plautz G E, Xu L, Felgner J H, Wheeler C J, Felgner P, Gao X, Huang L, Gordon D, Nabel G J, Nabel E G. Hum Gene Ther. 1993;4:781–788. - PubMed
-
- Nabel G J, Chang A E, Nabel E G, Plautz G E, Ensminger W, Fox B A, Felgner P, Shu S, Cho K. Hum Gene Ther. 1994;5:57–77. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

