Specialized membrane domains for water transport in glial cells: high-resolution immunogold cytochemistry of aquaporin-4 in rat brain
- PMID: 8987746
- PMCID: PMC6793699
- DOI: 10.1523/JNEUROSCI.17-01-00171.1997
Specialized membrane domains for water transport in glial cells: high-resolution immunogold cytochemistry of aquaporin-4 in rat brain
Abstract
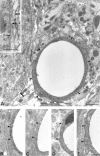
Membrane water transport is critically involved in brain volume homeostasis and in the pathogenesis of brain edema. The cDNA encoding aquaporin-4 (AQP4) water channel protein was recently isolated from rat brain. We used immunocytochemistry and high-resolution immunogold electron microscopy to identify the cells and membrane domains that mediate water flux through AQP4. The AQP4 protein is abundant in glial cells bordering the subarachnoidal space, ventricles, and blood vessels. AQP4 is also abundant in osmosensory areas, including the supraoptic nucleus and subfornical organ. Immunogold analysis demonstrated that AQP4 is restricted to glial membranes and to subpopulations of ependymal cells. AQP4 is particularly strongly expressed in glial membranes that are in direct contact with capillaries and pia. The highly polarized AQP4 expression indicates that these cells are equipped with specific membrane domains that are specialized for water transport, thereby mediating the flow of water between glial cells and the cavities filled with CSF and the intravascular space.
Figures





References
-
- Agre P, Preston GM, Smith BL, Jung JS, Raina S, Moon C, Guggino WB, Nielsen S. Aquaporin CHIP: the archetypal molecular water channel. Am J Physiol. 1993;265:F463–F476. - PubMed
-
- Armstrong WE, Schoeler J, McNeill TH. Immunocytochemical, Golgi and electron microscopic characterization of putative dendrites in the ventral glial lamina of the rat supraoptic nucleus. Neuroscience. 1982;7:679–694. - PubMed
-
- Chaudhry FA, Lehre KP, van Lookeren Campagne M, Ottersen OP, Danbolt NC, Storm-Mathisen J. Glutamate transporters in glial plasma membranes: highly differentiated localizations revealed by quantitative ultrastructural immunocytochemistry. Neuron. 1995;15:711–720. - PubMed
-
- Ecelbarger C, Terris J, Frindt G, Echevarria M, Marples D, Nielsen S, Knepper MA. Aquaporin-3 water channel localization and regulation in rat kidney. Am J Physiol. 1995;269:F663–F672. - PubMed
-
- Fitzsimons JT. Physiology and pathophysiology of thirst and sodium appetite. In: Seldin DW, Giebisch G, editors. The kidney: physiology and pathophysiology. Raven; New York: 1992. pp. 1615–1648.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
