Costorage and corelease of modulatory peptide cotransmitters with partially antagonistic actions on the accessory radula closer muscle of Aplysia californica
- PMID: 8987834
- PMCID: PMC6579219
- DOI: 10.1523/JNEUROSCI.16-24-08092.1996
Costorage and corelease of modulatory peptide cotransmitters with partially antagonistic actions on the accessory radula closer muscle of Aplysia californica
Abstract
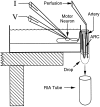
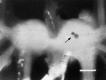
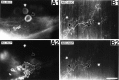
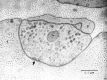
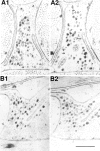
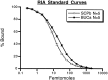
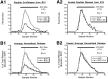
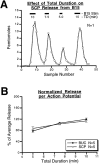
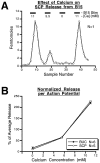
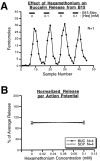
Many neurons that contain a classical neurotransmitter also contain modulatory peptides, but it has been difficult to establish unequivocally that these peptides are functional cotransmitters. Here, we provide evidence for functional cotransmission in a neuromuscular system of Aplysia. Using immunocytochemical techniques, we localize members of two peptide families, the small cardioactive peptides (SCPs) and the buccalins (BUCs), to a single subset of dense-core vesicles in the terminals of the cholinergic motorneuron B15. We describe a new preparation and method for the direct detection of released peptides and show that the SCPs and BUCs are released when neuron B15 is intracellularly stimulated. Consistent with their subcellular localization, the SCPs and BUCs are released in a stoichiometric ratio that is constant across conditions that change the absolute amount of peptides released. Peptide release is calcium-dependent but does not require muscle contractions. Thus, the release cannot be attributed to a displacement of peptides that may be present in the extracellular space. In previous studies, we characterized the physiological firing patterns of neuron B15. Here, we simulate these firing patterns and show that peptide release occurs. Additionally, we find that significant quantities of material are released under behaviorally relevant conditions. We find that concentrations of released peptides in the muscle are in the concentration range in which exogenously applied peptides exert characterized modulatory actions on muscle contractions. Together, our findings provide strong support for the hypothesis that peptides contained in neuron B15 are functional cotransmitters.
Figures

References
-
- Adams ME, O’Shea M. Peptide cotransmitter at a neuromuscular junction. Science. 1983;221:286–289. - PubMed
-
- Brezina V, Bank B, Cropper EC, Rosen S, Vilim FS, Kupfermann I, Weiss KR. Nine members of the myomodulin family of peptide cotransmitters at the B16-ARC neuromuscular junction of Aplysia. J Neurophysiol. 1995;74:54–72. - PubMed
-
- Cohen JL, Weiss KR, Kupfermann I. Motor control of buccal muscles in Aplysia. J Neurophysiol. 1978;41:157–180. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
