Actions of the ORL1 receptor ligand nociceptin on membrane properties of rat periaqueductal gray neurons in vitro
- PMID: 8994054
- PMCID: PMC6573185
- DOI: 10.1523/JNEUROSCI.17-03-00996.1997
Actions of the ORL1 receptor ligand nociceptin on membrane properties of rat periaqueductal gray neurons in vitro
Abstract
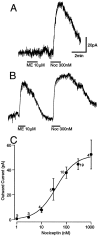
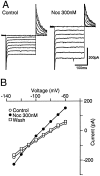
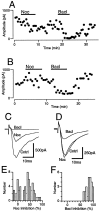
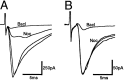
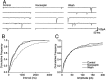
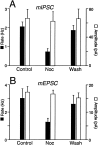
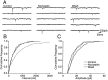
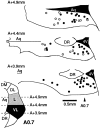
The actions of the endogenous ORL1-receptor ligand nociceptin on the membrane properties and synaptic currents in rat periaqueductal gray (PAG) neurons were examined by the use of whole-cell patch-clamp recording in brain slices. Nociceptin produced an outward current in all neurons tested, with an EC50 of 39 +/- 7 nM. The outward current was unaffected by naloxone. Outward currents reversed polarity at -110 +/- 3 mV in 2.5 mM extracellular potassium, and the reversal potential increased when the extracellular potassium concentration was raised (slope = 66.3 mV/log[K+]o mM). Thus, the nociceptin-induced outward current was attributable to an increased K+ conductance. Nociceptin inhibited evoked fast GABAergic (IP-SCs) and glutamatergic (EPSCs) postsynaptic currents and increased paired-pulse facilitation in a subpopulation of PAG neurons. Nociceptin inhibited evoked IPSCs and EPSCs in approximately 50% of neurons throughout the PAG, except in the ventrolateral PAG, where nociceptin inhibited evoked IPSCs in most neurons. Nociceptin decreased the frequency of spontaneous miniature postsynaptic currents (mIPSCs and mEPSCs) in a subpopulation of PAG neurons but had no effect on their amplitude distributions. Thus, nociceptin had a presynaptic inhibitory effect on transmitter release. These findings suggest that nociceptin, via its pre- and postsynaptic actions, has the potential to modulate the analgesic, behavioral, and autonomic functions of the PAG.
Figures
Similar articles
-
Presynaptic inhibitory action of opioids on synaptic transmission in the rat periaqueductal grey in vitro.J Physiol. 1997 Jan 15;498 ( Pt 2)(Pt 2):463-72. doi: 10.1113/jphysiol.1997.sp021872. J Physiol. 1997. PMID: 9032693 Free PMC article.
-
Actions of nociceptin/orphanin FQ and other prepronociceptin products on rat rostral ventromedial medulla neurons in vitro.J Physiol. 2001 Aug 1;534(Pt 3):849-59. doi: 10.1111/j.1469-7793.2001.00849.x. J Physiol. 2001. PMID: 11483714 Free PMC article.
-
Nociceptin receptor coupling to a potassium conductance in rat locus coeruleus neurones in vitro.Br J Pharmacol. 1996 Dec;119(8):1614-8. doi: 10.1111/j.1476-5381.1996.tb16080.x. Br J Pharmacol. 1996. PMID: 8982509 Free PMC article.
-
Inhibition by opioids acting on mu-receptors of GABAergic and glutamatergic postsynaptic potentials in single rat periaqueductal gray neurones in vitro.Br J Pharmacol. 1994 Sep;113(1):303-9. doi: 10.1111/j.1476-5381.1994.tb16209.x. Br J Pharmacol. 1994. PMID: 7812626 Free PMC article.
-
Pharmacological characterization of the nociceptin receptor, ORL1. Insight from the inward rectifier activation in the periaqueductal gray.J Biomed Sci. 2000 May-Jun;7(3):232-40. doi: 10.1007/BF02255471. J Biomed Sci. 2000. PMID: 10810242 Review.
Cited by
-
Estradiol negatively modulates the pleiotropic actions of orphanin FQ/nociceptin at proopiomelanocortin synapses.Neuroendocrinology. 2013;98(1):60-72. doi: 10.1159/000351868. Epub 2013 Jul 2. Neuroendocrinology. 2013. PMID: 23735696 Free PMC article.
-
The vital role of arcuate nociceptin/orphanin FQ neurones in mounting an oestradiol-dependent adaptive response to negative energy balance via inhibition of nearby proopiomelanocortin neurones.J Physiol. 2022 Nov;600(22):4939-4961. doi: 10.1113/JP283378. Epub 2022 Oct 28. J Physiol. 2022. PMID: 36217719 Free PMC article.
-
Induction of delta-opioid receptor function in the midbrain after chronic morphine treatment.J Neurosci. 2005 Mar 23;25(12):3192-8. doi: 10.1523/JNEUROSCI.4585-04.2005. J Neurosci. 2005. PMID: 15788776 Free PMC article.
-
Altered processing of pro-orphanin FQ/nociceptin and pro-opiomelanocortin-derived peptides in the brains of mice expressing defective prohormone convertase 2.J Neurosci. 2001 Aug 15;21(16):5864-70. doi: 10.1523/JNEUROSCI.21-16-05864.2001. J Neurosci. 2001. PMID: 11487609 Free PMC article.
-
[Phe1psi(CH2-NH)Gly2]nociceptin-(1 - 13)-NH2 activation of an inward rectifier as a partial agonist of ORL1 receptors in rat periaqueductal gray.Br J Pharmacol. 1999 Sep;128(1):103-7. doi: 10.1038/sj.bjp.0702746. Br J Pharmacol. 1999. PMID: 10498840 Free PMC article.
References
-
- Anton B, Fein J, To T, Li X, Silberstein L, Evans CJ. Immunohistochemical localization of ORL1 in the central nervous system of the rat. J Comp Neurol. 1996;368:229–251. - PubMed
-
- Bandler R, Shipley MT. Columnar organisation in the midbrain periaqueductal gray: modules for emotional expression? Trends Neurosci. 1994;17:379–389. - PubMed
-
- Barry PH. JPCalc, a software package for calculating liquid junction potential corrections in patch-clamp, intracellular, epithelial, and bilayer measurements and for correcting junction potential measurements. J Neurosci Methods. 1994;51:107–116. - PubMed
-
- Basbaum AI, Fields HL. Endogenous pain control systems: brainstem spinal pathways and endorphin circuitry. Annu Rev Neurosci. 1984;7:309–338. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
