Phospholipase C-beta1 is present in the botrysome, an intermediate compartment-like organelle, and Is regulated by visual experience in cat visual cortex
- PMID: 9006988
- PMCID: PMC6793723
- DOI: 10.1523/JNEUROSCI.17-04-01471.1997
Phospholipase C-beta1 is present in the botrysome, an intermediate compartment-like organelle, and Is regulated by visual experience in cat visual cortex
Abstract
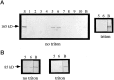
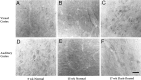
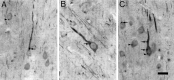
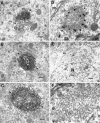
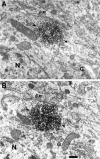
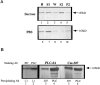
Monoclonal antibody Cat-307 identifies a 165 kDa neuronal protein expressed in the cat visual cortex during the period of sensitivity to alterations in visual experience (). Dark-rearing, which prolongs the sensitive period, also prolongs the expression of the Cat-307 protein. The Cat-307 protein localizes to an organelle, here called the botrysome (from the Greek botrys, cluster of grapes), that is located between the endoplasmic reticulum (ER) and Golgi apparatus. The botrysome is composed of small ring-shaped profiles with electron-dense coats. The size and morphology of the rings and their coats are similar to those described for ER to Golgi transport vesicles. Biochemically, the Cat-307 protein cofractionates with microsomes and partitions with subunits of the coatomer proteins that coat ER-to-Golgi transport vesicles. Partial amino acid sequencing reveals that the Cat-307 protein is phospholipase C-beta1, the G-protein-dependent phosphodiesterase that hydrolyses phosphatidylinositol 4,5 biphosphate into inositol 1,4,5 triphosphate and diacylglycerol after the stimulation of a variety of neurotransmitter receptors at the cell surface. These results suggest a role for phospholipase C-beta1 and the botrysome in developmental plasticity and provide a possible link between receptor activation at the cell surface and protein transport during neuronal development.
Figures

Similar articles
-
Structure of the Secretory Compartments in Goblet Cells in the Colon and Small Intestine.Cells. 2025 Jul 31;14(15):1185. doi: 10.3390/cells14151185. Cells. 2025. PMID: 40801617 Free PMC article.
-
Peripuberty Is a Sensitive Period for Prefrontal Parvalbumin Interneuron Activity to Impact Adult Cognitive Flexibility.Dev Neurosci. 2025;47(2):127-138. doi: 10.1159/000539584. Epub 2024 Jun 3. Dev Neurosci. 2025. PMID: 38830346 Free PMC article.
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Nicotine receptor partial agonists for smoking cessation.Cochrane Database Syst Rev. 2016 May 9;2016(5):CD006103. doi: 10.1002/14651858.CD006103.pub7. Cochrane Database Syst Rev. 2016. Update in: Cochrane Database Syst Rev. 2023 May 5;5:CD006103. doi: 10.1002/14651858.CD006103.pub8. PMID: 27158893 Free PMC article. Updated.
Cited by
-
Cellular and subcellular localization of PKMζ.Philos Trans R Soc Lond B Biol Sci. 2013 Dec 2;369(1633):20130140. doi: 10.1098/rstb.2013.0140. Print 2014 Jan 5. Philos Trans R Soc Lond B Biol Sci. 2013. PMID: 24298142 Free PMC article.
-
The unipolar brush cell: a remarkable neuron finally receiving deserved attention.Brain Res Rev. 2011 Jan 7;66(1-2):220-45. doi: 10.1016/j.brainresrev.2010.10.001. Epub 2010 Nov 5. Brain Res Rev. 2011. PMID: 20937306 Free PMC article. Review.
-
Immunohistochemical localization of huntingtin-associated protein 1 in endocrine system of the rat.J Histochem Cytochem. 2005 Dec;53(12):1517-24. doi: 10.1369/jhc.5A6662.2005. Epub 2005 Aug 8. J Histochem Cytochem. 2005. PMID: 16087704 Free PMC article.
-
The cellular and subcellular localization of huntingtin-associated protein 1 (HAP1): comparison with huntingtin in rat and human.J Neurosci. 1998 Oct 1;18(19):7674-86. doi: 10.1523/JNEUROSCI.18-19-07674.1998. J Neurosci. 1998. PMID: 9742138 Free PMC article.
-
Commentary on "E. Mugnaini and A. Floris, the unipolar brush cell: a neglected neuron of the mammalian cerebellar cortex. J Comp Neurol, 339:174-180, 1994".Cerebellum. 2015 Oct;14(5):484-6. doi: 10.1007/s12311-015-0660-1. Cerebellum. 2015. PMID: 25822473 No abstract available.
References
-
- Antonini A, Stryker MP. Rapid remodeling of axonal arbors in the visual cortex. Science. 1993a;260:1819–1821. - PubMed
-
- Bashir ZI, Collingridge GL. Synaptic plasticity: long-term potentiation in the hippocampus. Curr Opin Neurobiol. 1992;2:328–335. - PubMed
-
- Bashir ZI, Bortolotto ZA, Davies CH, Berretta N, Irving AJ, Seal AJ, Henley JM, Jane DE, Watkins JC, Collingridge GL. Induction of LTP in the hippocampus needs synaptic activation of glutamate metabotropic receptors. Nature. 1993;363:347–350. - PubMed
-
- Bauerfiend FR, Huttner WB. Biogenesis of constitutive secretory vesicles, secretory granules and synaptic vesicles. Curr Opin Cell Biol. 1993;5:628–635. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
