Polyphosphate kinase as a nucleoside diphosphate kinase in Escherichia coli and Pseudomonas aeruginosa
- PMID: 9012801
- PMCID: PMC19530
- DOI: 10.1073/pnas.94.2.439
Polyphosphate kinase as a nucleoside diphosphate kinase in Escherichia coli and Pseudomonas aeruginosa
Abstract
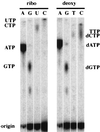
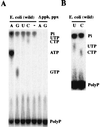
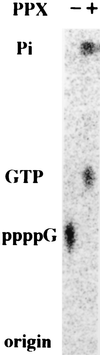
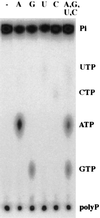
Generation of a wide variety of nucleoside (and deoxynucleoside) triphosphates (NTPs) from their cognate nucleoside diphosphates (NDPs) is of critical importance in virtually every aspect of cellular life. Their function is fulfilled largely by the ubiquitous and potent nucleoside diphosphate kinase (NDK), most commonly using ATP as the donor. Considerable interest is attached to the consequence to a cell in which the NDK activity becomes deficient or over-abundant. We have discovered an additional and possibly auxiliary NDK-like activity in the capacity of polyphosphate kinase (PPK) to use inorganic polyphosphate as the donor in place of ATP, thereby converting GDP and other NDPs to NTPs. This reaction was observed with the PPK activity present in crude membrane fractions from Escherichia coli and Pseudomonas aeruginosa as well as with the purified PPK from E. coli; the activity was absent from the membrane fractions obtained from E. coli mutants lacking the ppk gene. The order of substrate specificity for PPK was: ADP > GDP > UDP, CDP; activity with ADP was 2-60 times greater than with GDP, depending on the reaction condition. Although the transfer of a phosphate from polyphosphate to GDP by PPK to produce GTP was the predominant reaction, the enzyme also transferred a pyrophosphate group to GDP to form the linear guanosine 5' tetraphosphate.
Figures

References
-
- Biggs J, Hersperger E, Steeg P S, Liotta L A, Shearn A. Cell. 1990;63:933–940. - PubMed
-
- Munoz-Dorado J, Inouye M, Inouye S. J Biol Chem. 1990;265:2702–2706. - PubMed
-
- Wallet V, Mutzel R, Troll H, Barzu O, Wurster B, Veron M, Lacombe M L. J Natl Cancer Inst. 1990;82:1199–1202. - PubMed
-
- Okabe-Kado J, Kasukabe T, Hozumi M, Honma Y, Kimura N, Baba H, Urano T, Shiku H. FEBS Lett. 1995;363:311–315. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

