Targeted disruption of the mouse alpha A-crystallin gene induces cataract and cytoplasmic inclusion bodies containing the small heat shock protein alpha B-crystallin
- PMID: 9023351
- PMCID: PMC19608
- DOI: 10.1073/pnas.94.3.884
Targeted disruption of the mouse alpha A-crystallin gene induces cataract and cytoplasmic inclusion bodies containing the small heat shock protein alpha B-crystallin
Abstract
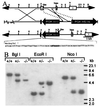
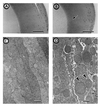
alpha A-crystallin (alpha A) and alpha B-crystallin (alpha B) are among the predominant proteins of the vertebrate eye lens. In vitro, the alpha-crystallins, which are isolated together as a high molecular mass aggregate, exhibit a number of properties, the most interesting of which is their ability to function as molecular chaperones for other proteins. Here we begin to examine the in vivo functions of alpha-crystallin by generating mice with a targeted disruption of the alpha A gene. Mice that are homozygous for the disrupted allele produce no detectable alpha A in their lenses, based on protein gel electrophoresis and immunoblot analysis. Initially, the alpha A-deficient lenses appear structurally normal, but they are smaller than the lenses of wild-type littermates. alpha A-/- lenses develop an opacification that starts in the nucleus and progresses to a general opacification with age. Light and transmission electron microscopy reveal the presence of dense inclusion bodies in the central lens fiber cells. The inclusions react strongly with antibodies to alpha B but not significantly with antibodies to beta- or gamma-crystallins. In addition, immunoblot analyses demonstrate that a significant portion of the alpha B in alpha A-/- lenses shifts into the insoluble fraction. These studies suggest that alpha A is essential for maintaining lens transparency, possibly by ensuring that alpha B or proteins closely associated with this small heat shock protein remain soluble.
Figures




References
-
- Benedek G B. Appl Opt. 1971;10:459–473. - PubMed
-
- Piatigorsky J. Differentiation (Berlin) 1981;19:134–153. - PubMed
-
- de Jong W W. In: Molecular and Cellular Biology of the Eye Lens. Bloemendal H, editor. New York: Wiley; 1981. pp. 221–278.
-
- Kato K, Shinohara H, Goto S, Inaguma Y, Morishita R, Asano T. J Biol Chem. 1992;267:7718–7725. - PubMed
-
- Srinivasan A N, Nagineni C N, Bhat S P. J Biol Chem. 1992;267:23337–23341. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

