Suppression of apoptosis by nitric oxide via inhibition of interleukin-1beta-converting enzyme (ICE)-like and cysteine protease protein (CPP)-32-like proteases
- PMID: 9034139
- PMCID: PMC2196141
- DOI: 10.1084/jem.185.4.601
Suppression of apoptosis by nitric oxide via inhibition of interleukin-1beta-converting enzyme (ICE)-like and cysteine protease protein (CPP)-32-like proteases
Abstract
Physiological levels of shear stress alter the genetic program of cultured endothelial cells and are associated with reduced cellular turnover rates and formation of atherosclerotic lesions in vivo. To test the hypothesis that shear stress (15 dynes/cm2) interferes with programmed cell death, apoptosis was induced in human umbilical venous cells (HUVEC) by tumor necrosis factor-alpha (TNF-alpha). Apoptosis was quantified by ELISA specific for histone-associated DNA-fragments and confirmed by demonstrating the specific pattern of internucleosomal DNA-fragmentation. TNF-alpha (300 U/ml) mediated increase of DNA-fragmentation was completely abrogated by shear stress (446 +/- 121% versus 57 +/- 11%, P <0.05). This anti-apoptotic activity of shear stress decreased after pharmacological inhibition of endogenous nitric oxide (NO)-synthase by NG-monomethyl-L-arginine and was completely reproduced by exogenous NO-donors. The activation of interleukin-1beta-converting enzyme (ICE)-like and cysteine protease protein (CPP)-32-like cysteine proteases was required to mediate TNF-alpha-induced apoptosis of HUVEC. Endothelial-derived nitric oxide (NO) as well as exogenous NO donors inhibited TNF-alpha-induced cysteine protease activation. Inhibition of CPP-32 enzyme activity was due to specific S-nitrosylation of Cys 163, a functionally essential amino acid conserved among ICE/CPP-32-like proteases. Thus, we propose that shear stress-mediated NO formation interferes with cell death signal transduction and may contribute to endothelial cell integrity by inhibition of apoptosis.
Figures
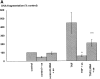


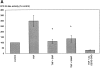




References
-
- Ku DN, Giddens DP, Zarins CK, Glagov S. Pulsatile flow and atherosclerosis in the human carotid bifurcation. Positive correlation between plaque location and low oscillating shear stress. Arteriosclerosis. 1985;5:293–302. - PubMed
-
- Caplan BA, Schwartz CJ. Increased endothelial cell turnover in areas of in vivo Evans Blue uptake in the pig aorta. Atherosclerosis. 1973;17:401–417. - PubMed
-
- Cohen JJ. Apoptosis. Immunol Today. 1993;14:126–130. - PubMed
-
- Steller H. Mechanisms and genes of cellular suicide. Science (Wash DC) 1995;267:1445–1449. - PubMed
-
- Kumar S. ICE-like proteases in apoptosis. Trends Biochem Sci. 1995;20:198–202. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

