Dihydropyridine action on voltage-dependent potassium channels expressed in Xenopus oocytes
- PMID: 9041446
- PMCID: PMC2220064
- DOI: 10.1085/jgp.109.2.169
Dihydropyridine action on voltage-dependent potassium channels expressed in Xenopus oocytes
Abstract
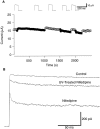
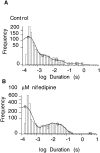
Dihydropyridines (DHPs) are well known for their effects on L-typed voltage-dependent Ca2+ channels, However, these drugs also affect other voltage-dependent ion channels, including Shaker K+ channels. We examined the effects of DHPs on the Shaker K+ channels expressed in Xenopus oocytes. Intracellular applications of DHPs quickly and reversibly induced apparent inactivation in the Shaker K+ mutant channels with disrupted N- and C-type inactivation. We found that DHPs interact with the open state of the channel as evidenced by the decreased mean open time. The DHPs effects are voltage-dependent, becoming more effective with hyperpolarization. A model which involves binding of two DHP molecules to the channel is consistent with the results obtained in our experiments.
Figures









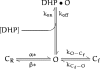



Similar articles
-
Differential blocking action of dihydropyridine Ca2+ antagonists on a T-type Ca2+ channel (alpha1G) expressed in Xenopus oocytes.J Cardiovasc Pharmacol. 2005 Mar;45(3):241-6. doi: 10.1097/01.fjc.0000154374.88283.15. J Cardiovasc Pharmacol. 2005. PMID: 15725949
-
Determinants of voltage-dependent gating and open-state stability in the S5 segment of Shaker potassium channels.J Gen Physiol. 1999 Aug;114(2):215-42. doi: 10.1085/jgp.114.2.215. J Gen Physiol. 1999. PMID: 10435999 Free PMC article.
-
Voltage-insensitive gating after charge-neutralizing mutations in the S4 segment of Shaker channels.J Gen Physiol. 1999 Jan;113(1):139-51. doi: 10.1085/jgp.113.1.139. J Gen Physiol. 1999. PMID: 9874694 Free PMC article.
-
Fused 1,4-dihydropyridines as potential calcium modulatory compounds.Mini Rev Med Chem. 2006 Jul;6(7):747-55. doi: 10.2174/138955706777698606. Mini Rev Med Chem. 2006. PMID: 16842124 Review.
-
Functional and molecular aspects of voltage-gated K+ channel beta subunits.Ann N Y Acad Sci. 1999 Apr 30;868:344-55. doi: 10.1111/j.1749-6632.1999.tb11296.x. Ann N Y Acad Sci. 1999. PMID: 10414304 Review.
Cited by
-
Dual and opposing roles of presynaptic Ca2+ influx for spontaneous GABA release from rat medial preoptic nerve terminals.J Physiol. 2002 Jul 1;542(Pt 1):131-46. doi: 10.1113/jphysiol.2001.015610. J Physiol. 2002. PMID: 12096057 Free PMC article.
-
Histidine(118) in the S2-S3 linker specifically controls activation of the KAT1 channel expressed in Xenopus oocytes.Biophys J. 2000 Mar;78(3):1255-69. doi: 10.1016/S0006-3495(00)76682-3. Biophys J. 2000. PMID: 10692314 Free PMC article.
-
Acceleration of P/C-type inactivation in voltage-gated K(+) channels by methionine oxidation.Biophys J. 2000 Jan;78(1):174-87. doi: 10.1016/S0006-3495(00)76583-0. Biophys J. 2000. PMID: 10620284 Free PMC article.
-
Mining of Ebola virus entry inhibitors identifies approved drugs as two-pore channel pore blockers.Biochim Biophys Acta Mol Cell Res. 2019 Jul;1866(7):1151-1161. doi: 10.1016/j.bbamcr.2018.10.022. Epub 2018 Nov 5. Biochim Biophys Acta Mol Cell Res. 2019. PMID: 30408544 Free PMC article.
-
Calcium-induced transitions between the spontaneous miniature outward and the transient outward currents in retinal amacrine cells.J Gen Physiol. 2002 Apr;119(4):373-88. doi: 10.1085/jgp.20028479. J Gen Physiol. 2002. PMID: 11929887 Free PMC article.
References
-
- Baukrowitz T, Yellen G. Use-dependent blockers and exit rate of the last ion from the multi-pore of a K+channel. Science (Wash DC) 1996;271:653–656. - PubMed
-
- Colquhoun, D., and A.G. Hawkes. 1995. A Q-matrix cookbook: how to write only one program to calculate the single-channel and macroscopic predictions for any kinetic mechanism. In Single-channel recording. Plenum, New York. 589–633.
-
- Demo SD, Yellen G. The inactivation gate of the Shaker K+channel behaves like an open-channel blocker. Neuron. 1991;7:743–753. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

