Development of multisensory neurons and multisensory integration in cat superior colliculus
- PMID: 9065504
- PMCID: PMC6573512
- DOI: 10.1523/JNEUROSCI.17-07-02429.1997
Development of multisensory neurons and multisensory integration in cat superior colliculus
Abstract
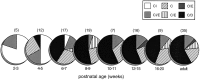
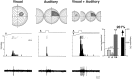
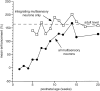
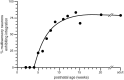
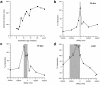
The development of multisensory neurons and multisensory integration was examined in the deep layers of the superior colliculus of kittens ranging in age from 3 to 135 d postnatal (dpn). Despite the high proportion of multisensory neurons in adult animals, no such neurons were found during the first 10 d of postnatal life. Rather, all sensory-responsive neurons were unimodal. The first multisensory neurons (somatosensory-auditory) were found at 12 dpn, and visually responsive multisensory neurons were not found until 20 dpn. Early multisensory neurons responded weakly to sensory stimuli, had long latencies, large receptive fields, and poorly developed response selectivities. Most surprising, however, was their inability to integrate combinations of sensory cues to produce significant response enhancement (or depression), a characteristic feature of the adult. Responses to combinations of sensory cues differed little from responses to their modality-specific components. At 28 dpn an abrupt physiological change was noted. Some multisensory neurons now integrated combinations of cross-modality cues and exhibited significant response enhancements when these cues were spatially coincident and response depressions when the cues were spatially disparate. During the next 2 months the incidence of multisensory neurons, and the proportion of these neurons capable of adult-like multisensory integration, gradually increased. Once multisensory integration appeared in a given neuron, its properties changed little with development. Even the youngest integrating neurons showed superadditive enhancements and spatial characteristics of multisensory integration that were indistinguishable from the adult. Nevertheless, neonatal and adult multisensory neurons differed in the manner in which they integrated temporally asynchronous stimuli, a distribution that may reflect the very different behavioral requirements at different ages. The possible maturational role of corticotectal projections in the abrupt gating of multisensory integration is discussed.
Figures







Similar articles
-
Representation and integration of multiple sensory inputs in primate superior colliculus.J Neurophysiol. 1996 Aug;76(2):1246-66. doi: 10.1152/jn.1996.76.2.1246. J Neurophysiol. 1996. PMID: 8871234
-
Spatial determinants of multisensory integration in cat superior colliculus neurons.J Neurophysiol. 1996 May;75(5):1843-57. doi: 10.1152/jn.1996.75.5.1843. J Neurophysiol. 1996. PMID: 8734584
-
Sensory and multisensory responses in the newborn monkey superior colliculus.J Neurosci. 2001 Nov 15;21(22):8886-94. doi: 10.1523/JNEUROSCI.21-22-08886.2001. J Neurosci. 2001. PMID: 11698600 Free PMC article.
-
Neural mechanisms for synthesizing sensory information and producing adaptive behaviors.Exp Brain Res. 1998 Nov;123(1-2):124-35. doi: 10.1007/s002210050553. Exp Brain Res. 1998. PMID: 9835401 Review.
-
Nonvisual influences on visual-information processing in the superior colliculus.Prog Brain Res. 2001;134:143-56. doi: 10.1016/s0079-6123(01)34011-6. Prog Brain Res. 2001. PMID: 11702540 Review.
Cited by
-
Multisensory Plasticity in Superior Colliculus Neurons is Mediated by Association Cortex.Cereb Cortex. 2016 Mar;26(3):1130-7. doi: 10.1093/cercor/bhu295. Epub 2014 Dec 31. Cereb Cortex. 2016. PMID: 25552270 Free PMC article.
-
Multisensory plasticity in adulthood: cross-modal experience enhances neuronal excitability and exposes silent inputs.J Neurophysiol. 2013 Jan;109(2):464-74. doi: 10.1152/jn.00739.2012. Epub 2012 Oct 31. J Neurophysiol. 2013. PMID: 23114212 Free PMC article.
-
The development of a dialogue between cortex and midbrain to integrate multisensory information.Exp Brain Res. 2005 Oct;166(3-4):305-15. doi: 10.1007/s00221-005-2372-0. Epub 2005 Jun 30. Exp Brain Res. 2005. PMID: 15988596 Review.
-
Cross-Modal Competition: The Default Computation for Multisensory Processing.J Neurosci. 2019 Feb 20;39(8):1374-1385. doi: 10.1523/JNEUROSCI.1806-18.2018. Epub 2018 Dec 20. J Neurosci. 2019. PMID: 30573648 Free PMC article.
-
Postnatal development of sound pressure transformations by the head and pinnae of the cat: monaural characteristics.J Acoust Soc Am. 2009 Feb;125(2):980-94. doi: 10.1121/1.3058630. J Acoust Soc Am. 2009. PMID: 19206874 Free PMC article.
References
-
- Baleydier C, Kahungu M, Mauguiere F. A crossed corticotectal projection from the lateral suprasylvian area in the cat. J Comp Neurol. 1983;214:344–351. - PubMed
-
- Berson DM. Cat lateral suprasylvian cortex: Y-cell inputs and corticotectal projections. J Neurophysiol. 1985;53:544–556. - PubMed
-
- Binns KE, Salt TE. Importance of NMDA receptors for multimodal integration in the deep layers of the cat superior colliculus. J Neurophysiol. 1996;75:920–930. - PubMed
-
- Clemo HR, Stein BE. Effects of cooling somatosensory cortex on response properties of tactile cells in superior colliculus. J Neurophysiol. 1986;55:1352–1368. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
