Chromatin degradation in differentiating fiber cells of the eye lens
- PMID: 9105035
- PMCID: PMC2139849
- DOI: 10.1083/jcb.137.1.37
Chromatin degradation in differentiating fiber cells of the eye lens
Abstract
During development, the lens of the eye becomes transparent, in part because of the elimination of nuclei and other organelles from the central lens fiber cells by an apoptotic-like mechanism. Using confocal microscopy we showed that, at the border of the organelle-free zone (OFZ), fiber cell nuclei became suddenly irregular in shape, with marginalized chromatin. Subsequently, holes appeared in the nuclear envelope and underlying laminae, and the nuclei collapsed into condensed, spherical structures. Nuclear remnants, containing DNA, histones, lamin B2, and fragments of nuclear membrane, were detected deep in the OFZ. We used in situ electrophoresis to demonstrate that fragmented DNA was present only in cells bordering the OFZ. Confocal microscopy of terminal deoxynucleotidyl transferase (TdT)-labeled lens slices confirmed that DNA fragmentation was a relatively late event in fiber differentiation, occurring after the loss of the nuclear membrane. DNA fragments with 3'-OH or 3'-PO(4) ends were not observed elsewhere in the lens under normal conditions, although they could be produced by pretreatment with DNase I or micrococcal nuclease, respectively. Dual labeling with TdT and an antibody against protein disulfide isomerase, an ER-resident protein, revealed a distinct spatial and temporal gap between the disappearance of ER and nuclear membranes and the onset of DNA degradation. Thus, fiber cell chromatin disassembly differs significantly from classical apoptosis, in both the sequence of events and the time course of the process. The fact that DNA degradation occurs only after the disappearance of mitochondrial, ER, and nuclear membranes suggests that damage to intracellular membranes may be an initiating event in nuclear breakdown.
Figures


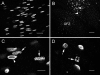


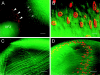

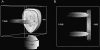

References
-
- Arruti C, Chaudun E, De Maria A, Courtois Y, Counis M-F. Characterization of eye-lens DNases: long term persistence of activity in post apoptotic lens fiber cells. Cell Death Differ. 1995;2:47–56. - PubMed
-
- Barry MA, Eastman A. Identification of deoxyribonuclease II as an endonuclease involved in apoptosis. Arch Biochem Biophys. 1993;300:440–450. - PubMed
-
- Bassnett S. Mitochondrial dynamics in differentiating fiber cells of the mammalian lens. Curr Eye Res. 1992;11:1227–1232. - PubMed
-
- Bassnett S. The fate of the Golgi apparatus and the endoplasmic reticulum during lens fiber cell differentiation. Invest Ophthalmol Visual Sci. 1995;36:1783–1803. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

