Receptor and G betagamma isoform-specific interactions with G protein-coupled receptor kinases
- PMID: 9122168
- PMCID: PMC20061
- DOI: 10.1073/pnas.94.6.2180
Receptor and G betagamma isoform-specific interactions with G protein-coupled receptor kinases
Abstract
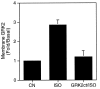
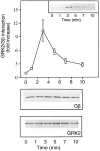
The G protein-coupled receptor (GPCR) kinases (GRKs) phosphorylate and desensitize agonist-occupied GPCRs. GRK2-mediated receptor phosphorylation is preceded by the agonist-dependent membrane association of this enzyme. Previous in vitro studies with purified proteins have suggested that this translocation may be mediated by the recruitment of GRK2 to the plasma membrane by its interaction with the free betagamma subunits of heterotrimeric G proteins (G betagamma). Here we demonstrate that this mechanism operates in intact cells and that specificity is imparted by the selective interaction of discrete pools of G betagamma with receptors and GRKs. Treatment of Cos-7 cells transiently overexpressing GRK2 with a beta-receptor agonist promotes a 3-fold increase in plasma membrane-associated GRK2. This translocation of GRK2 is inhibited by the carboxyl terminus of GRK2, a known G betagamma sequestrant. Furthermore, in cells overexpressing both GRK2 and G beta1 gamma2, activation of lysophosphatidic acid receptors leads to the rapid and transient formation of a GRK/G betagamma complex. That G betagamma specificity exists at the level of the GPCR and the GRK is indicated by the observation that a GRK2/G betagamma complex is formed after agonist occupancy of the lysophosphatidic acid and beta-adrenergic but not thrombin receptors. In contrast to GRK2, GRK3 forms a G betagamma complex after stimulation of all three GPCRs. This G betagamma binding specificity of the GRKs is also reflected at the level of the purified proteins. Thus the GRK2 carboxyl terminus binds G beta1 and G beta2 but not G beta3, while the GRK3 fusion protein binds all three G beta isoforms. This study provides a direct demonstration of a role for G betagamma in mediating the agonist-stimulated translocation of GRK2 and GRK3 in an intact cellular system and demonstrates isoform specificity in the interaction of these components.
Figures



Similar articles
-
Phosphatidylinositol 4,5-bisphosphate (PIP2)-enhanced G protein-coupled receptor kinase (GRK) activity. Location, structure, and regulation of the PIP2 binding site distinguishes the GRK subfamilies.J Biol Chem. 1996 Oct 4;271(40):24907-13. doi: 10.1074/jbc.271.40.24907. J Biol Chem. 1996. PMID: 8798768
-
Lipid-mediated regulation of G protein-coupled receptor kinases 2 and 3.J Biol Chem. 1995 Mar 17;270(11):5742-7. doi: 10.1074/jbc.270.11.5742. J Biol Chem. 1995. PMID: 7890702
-
Monoclonal antibodies reveal receptor specificity among G-protein-coupled receptor kinases.Proc Natl Acad Sci U S A. 1996 Jul 23;93(15):7649-54. doi: 10.1073/pnas.93.15.7649. Proc Natl Acad Sci U S A. 1996. PMID: 8755530 Free PMC article.
-
Protein kinases that phosphorylate activated G protein-coupled receptors.FASEB J. 1995 Feb;9(2):175-82. doi: 10.1096/fasebj.9.2.7781920. FASEB J. 1995. PMID: 7781920 Review.
-
GTP-binding-protein-coupled receptor kinases--two mechanistic models.Eur J Biochem. 1997 Sep 1;248(2):261-9. doi: 10.1111/j.1432-1033.1997.00261.x. Eur J Biochem. 1997. PMID: 9346277 Review.
Cited by
-
Noncanonical Roles of G Protein-coupled Receptor Kinases in Cardiovascular Signaling.J Cardiovasc Pharmacol. 2017 Sep;70(3):129-141. doi: 10.1097/FJC.0000000000000483. J Cardiovasc Pharmacol. 2017. PMID: 28328744 Free PMC article. Review.
-
Feedback regulation of G protein-coupled receptor signaling by GRKs and arrestins.Semin Cell Dev Biol. 2016 Feb;50:95-104. doi: 10.1016/j.semcdb.2015.12.015. Epub 2016 Jan 7. Semin Cell Dev Biol. 2016. PMID: 26773211 Free PMC article. Review.
-
Tumor necrosis factor-alpha prevents desensitization of Galphas-coupled receptors by regulating GRK2 association with the plasma membrane.Mol Pharmacol. 2006 Apr;69(4):1311-9. doi: 10.1124/mol.105.016857. Epub 2005 Dec 29. Mol Pharmacol. 2006. PMID: 16385076
-
Decreased GRK3 but not GRK2 expression in frontal cortex from bipolar disorder patients.Int J Neuropsychopharmacol. 2009 Jul;12(6):851-60. doi: 10.1017/S146114570900025X. Epub 2009 Apr 29. Int J Neuropsychopharmacol. 2009. Retraction in: Int J Neuropsychopharmacol. 2016 Dec 30;19(12):pyw085. doi: 10.1093/ijnp/pyw085. PMID: 19400979 Free PMC article. Retracted.
-
An integrated stochastic model of "inside-out" integrin activation and selective T-lymphocyte recruitment.Langmuir. 2012 Jan 31;28(4):2225-37. doi: 10.1021/la203803e. Epub 2012 Jan 4. Langmuir. 2012. PMID: 22149624 Free PMC article.
References
-
- Freedman N J, Lefkowitz R J. Recent Prog Horm Res. 1996;51:319–353. - PubMed
-
- Inglese J, Freedman N J, Koch W J, Lefkowitz R J. J Biol Chem. 1993;268:23735–23738. - PubMed
-
- Premont R T, Inglese J, Lefkowitz R J. FASEB J. 1995;9:175–182. - PubMed
-
- Pitcher J A, Inglese J, Higgins J B, Arriza J L, Casey P J, Kim C, Benovic J L, Kwatra M M, Caron M G, Lefkowitz R J. Science. 1992;257:1264–1267. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

