Evidence for a lipochaperonin: association of active protein-folding GroESL oligomers with lipids can stabilize membranes under heat shock conditions
- PMID: 9122170
- PMCID: PMC20063
- DOI: 10.1073/pnas.94.6.2192
Evidence for a lipochaperonin: association of active protein-folding GroESL oligomers with lipids can stabilize membranes under heat shock conditions
Abstract
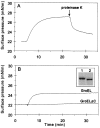
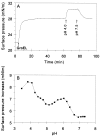
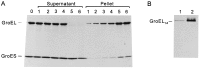
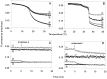
During heat shock, structural changes in proteins and membranes may lead to cell death. While GroE and other chaperone proteins are involved in the prevention of stress-induced protein aggregation and in the recovery of protein structures, a mechanism for short-term membrane stabilization during stress remains to be established. We found that GroEL chaperonin can associate with model lipid membranes. Binding was apparently governed by the composition and the physical state of the host bilayer. Limited proteolysis of GroEL oligomers by proteinase K, which removes selectively the conserved glycine- and methionine-rich C terminus, leaving the chaperonin oligomer intact, prevented chaperonin association with lipid membranes. GroEL increased the lipid order in the liquid crystalline state, yet remained functional as a protein-folding chaperonin. This suggests that, during stress, chaperonins can assume the functions of assisting the folding of both soluble and membrane-associated proteins while concomitantly stabilizing lipid membranes.
Figures

Similar articles
-
Stability of the asymmetric Escherichia coli chaperonin complex. Guanidine chloride causes rapid dissociation.J Biol Chem. 1995 Mar 10;270(10):5388-94. doi: 10.1074/jbc.270.10.5388. J Biol Chem. 1995. PMID: 7890652
-
The protein-folding activity of chaperonins correlates with the symmetric GroEL14(GroES7)2 heterooligomer.Proc Natl Acad Sci U S A. 1995 Dec 19;92(26):12021-5. doi: 10.1073/pnas.92.26.12021. Proc Natl Acad Sci U S A. 1995. PMID: 8618836 Free PMC article.
-
Reversible oligomerization and denaturation of the chaperonin GroES.Biochemistry. 1996 Apr 2;35(13):4079-83. doi: 10.1021/bi953087n. Biochemistry. 1996. PMID: 8672442
-
Reaction Cycle of Chaperonin GroEL via Symmetric "Football" Intermediate.J Mol Biol. 2015 Sep 11;427(18):2912-8. doi: 10.1016/j.jmb.2015.04.007. Epub 2015 Apr 18. J Mol Biol. 2015. PMID: 25900372 Review.
-
GroEL and the GroEL-GroES Complex.Subcell Biochem. 2017;83:483-504. doi: 10.1007/978-3-319-46503-6_17. Subcell Biochem. 2017. PMID: 28271487 Review.
Cited by
-
A Low Membrane Hsp70 Expression in Tumor Cells With Impaired Lactate Metabolism Mediates Radiosensitization by NVP-AUY922.Front Oncol. 2022 Apr 7;12:861266. doi: 10.3389/fonc.2022.861266. eCollection 2022. Front Oncol. 2022. PMID: 35463341 Free PMC article.
-
Neuroprotective effect of small heat shock protein, Hsp27, after acute and chronic alcohol administration.Cell Stress Chaperones. 2010 Nov;15(6):807-17. doi: 10.1007/s12192-010-0188-8. Epub 2010 May 12. Cell Stress Chaperones. 2010. PMID: 20461564 Free PMC article.
-
The Legionella pneumophila Chaperonin - An Unusual Multifunctional Protein in Unusual Locations.Front Microbiol. 2011 Jun 10;2:122. doi: 10.3389/fmicb.2011.00122. eCollection 2011. Front Microbiol. 2011. PMID: 21713066 Free PMC article.
-
In situ metabolomic- and transcriptomic-profiling of the host-associated cyanobacteria Prochloron and Acaryochloris marina.ISME J. 2018 Feb;12(2):556-567. doi: 10.1038/ismej.2017.192. Epub 2017 Oct 31. ISME J. 2018. PMID: 29087375 Free PMC article.
-
Mechanisms of heat shock response in mammals.Cell Mol Life Sci. 2013 Nov;70(22):4229-41. doi: 10.1007/s00018-013-1348-7. Epub 2013 Apr 30. Cell Mol Life Sci. 2013. PMID: 23633190 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

