Dynamic control of location-specific information in tactile cutaneous reflexes from the foot during human walking
- PMID: 9133399
- PMCID: PMC6573668
- DOI: 10.1523/JNEUROSCI.17-10-03804.1997
Dynamic control of location-specific information in tactile cutaneous reflexes from the foot during human walking
Abstract
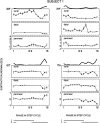
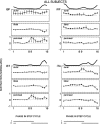
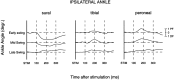
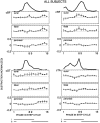
The purpose of the present study was to determine whether tactile cutaneous reflexes from the skin of the foot contain location-specific information during human walking. Muscular responses to non-nociceptive electrical stimulation of the sural, posterior tibial, and superficial peroneal nerves, each supplying a different skin area of the foot, were studied in both legs during walking on a treadmill. For all three nerves the major responses in all muscles were observed at a similar latency of approximately 80-85 msec. In the ipsilateral leg these reflex responses and their phase-dependent modulation were highly nerve-specific. During most of the stance phase, for example, the peroneal and tibial nerves generally evoked small responses in the biceps femoris muscle. In contrast, during late swing large facilitations generally occurred for the peroneal nerve, whereas suppressions were observed for the tibial nerve. In the contralateral leg the reflex responses for the three nerves were less distinct, although some nerve specificity was observed for individual subjects. It is concluded that non-nociceptive stimulation of the sural, posterior tibial, and superficial peroneal nerves each evokes distinct reflex responses, indicating the presence of location-specific information from the skin of the foot in cutaneous reflexes during human walking. It will be argued that differentially controlled reflex pathways can account for the differences in the phase-dependent reflex modulation patterns of the three nerves, which points to the dynamic control of this information during the course of a step cycle.
Figures


References
-
- Abraham LD, Marks WB, Loeb GE. The distal hindlimb musculature of the cat. Cutaneous reflexes during locomotion. Exp Brain Res. 1985;58:594–603. - PubMed
-
- Aniss AM, Gandevia SC, Burke D. Reflex changes in muscle spindle discharge during a voluntary contraction. J Neurophysiol. 1988;59:908–921. - PubMed
-
- Aniss AM, Gandevia SC, Burke D. Reflex responses in active muscles elicited by stimulation of low-threshold afferents from the human foot. J Neurophysiol. 1992;67:1375–1384. - PubMed
-
- Baker SN, Lemon RN. Non-linear summation of responses in averages of rectified EMG. J Neurosci Methods. 1995;59:175–181. - PubMed
-
- Buford JA, Smith JL. Adaptive control for backward quadrupedal walking. III. Stumbling corrective reactions and cutaneous reflex sensitivity. J Neurophysiol. 1993;70:1102–1114. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
