Cyp7b, a novel brain cytochrome P450, catalyzes the synthesis of neurosteroids 7alpha-hydroxy dehydroepiandrosterone and 7alpha-hydroxy pregnenolone
- PMID: 9144166
- PMCID: PMC24607
- DOI: 10.1073/pnas.94.10.4925
Cyp7b, a novel brain cytochrome P450, catalyzes the synthesis of neurosteroids 7alpha-hydroxy dehydroepiandrosterone and 7alpha-hydroxy pregnenolone
Abstract
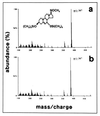
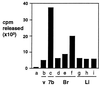
Steroids produced locally in brain (neurosteroids), including dehydroepiandrosterone (DHEA), influence cognition and behavior. We previously described a novel cytochrome P450, Cyp7b, strongly expressed in rat and mouse brain, particularly in hippocampus. Cyp7b is most similar to steroidogenic P450s and potentially could play a role in neurosteroid metabolism. To examine the catalytic activity of the enzyme mouse Cyp7b cDNA was introduced into a vaccinia virus vector. Extracts from cells infected with the recombinant showed NADPH-dependent conversion of DHEA (Km, 13.6 microM) and pregnenolone (Km, 4.0 microM) to slower migrating forms on thin layer chromatography. The expressed enzyme was less active against 25-hydroxycholesterol, 17beta-estradiol and 5alpha-androstane-3beta,17beta-diol, with low to undetectable activity against progesterone, corticosterone, and testosterone. On gas chromatography and mass spectrometry of the Cyp7b metabolite of DHEA the retention time and fragmentation patterns were identical to those obtained with authentic 7alpha-hydroxy DHEA. The reaction product also comigrated on thin layer chromatography with 7alpha-hydroxy DHEA but not with 7beta-hydroxy DHEA; when [7alpha-3H]pregnenolone was incubated with Cyp7b extracts the extent of release of radioactivity into the medium suggested that hydroxylation was preferentially at the 7alpha position. Brain extracts also efficiently liberated tritium from [7alpha-3H]pregnenolone and converted DHEA to a product with a chromatographic mobility indistinguishable from 7alpha-hydroxy DHEA. We conclude that Cyp7b is a 7alpha-hydroxylase participating in the synthesis, in brain, of neurosteroids 7alpha-hydroxy DHEA, and 7alpha-hydroxy pregnenolone.
Figures


References
-
- Robel P, Bourreau E, Corpéchot C, Dang D C, Halberg F, Clarke C, Haug M, Schlegel M L, Synguelakis M, Vourch C, Baulieu E E. J Steroid Biochem. 1987;27:649–655. - PubMed
-
- Robel P, Young J, Corpéchot C, Mayo W, Perché F, Haug M, Simon H, Baulieu E E. J Steroid Biochem Mol Biol. 1995;53:355–360. - PubMed
-
- Robel P, Baulieu E E. In: Neurobiological Effects of Sex Steroid Hormones. Micevych P E, Hammer R P, editors. Cambridge: Cambridge Univ. Press; 1995. pp. 281–296.
-
- Flood J F, Roberts E. Brain Res. 1988;448:178–181. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

