Spermine inhibits proinflammatory cytokine synthesis in human mononuclear cells: a counterregulatory mechanism that restrains the immune response
- PMID: 9151701
- PMCID: PMC2196317
- DOI: 10.1084/jem.185.10.1759
Spermine inhibits proinflammatory cytokine synthesis in human mononuclear cells: a counterregulatory mechanism that restrains the immune response
Abstract
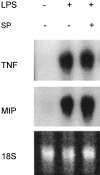
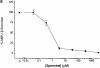
The local production of proinflammatory cytokines mediates the host response to inflammation, infection, and injury, whereas an overexpression of these mediators can injure or kill the host. Recently, we identified a class of multivalent guanylhydrazone compounds that are effective inhibitors of proinflammatory cytokine synthesis in monocytes/macrophages. The structure of one such cationic molecule suggested a molecular mimicry with spermine, a ubiquitous endogenous biogenic amine that increases significantly at sites of inflammation and infection. Here, we addressed the hypothesis that spermine might counterregulate the innate immune response by downregulating the synthesis of potentially injurious cytokines. When spermine was added to cultures of human peripheral blood mononuclear cells stimulated with lipopolysaccharide (LPS), it effectively inhibited the synthesis of the proinflammatory cytokines tumor necrosis factor (TNF), interleukin-1 (IL-1), IL-6, MIP-1alpha, and MIP-1beta. The inhibition of cytokine synthesis was specific and reversible, with significant inhibition of TNF synthesis occurring even when spermine was added after LPS. The mechanism of spermine-mediated cytokine suppression was posttranscriptional and independent of polyamine oxidase activity. Local administration of spermine in vivo protected mice against the development of acute footpad inflammation induced by carrageenan. These results identify a distinct molecular counterregulatory role for spermine in downregulating the monocyte proinflammatory cytokine response.
Figures











References
-
- Pfeffer K, Matsuyama T, Kundig TM, Wakeham A, Kishihara K, Shahinian A, Wiegmann K, Ohashi PS, Kronke M, Mak TW. Mice deficient for the 55 kd tumor necrosis factor receptor are resistant to endotoxic shock, yet succumb to L. monocytogenesinfection. Cell. 1993;73:457–467. - PubMed
-
- Tracey KJ, Beutler B, Lowry SF, Merryweather J, Wolpe S, Milsark IW, Hariri RJ, Fahey TF, III, Zentella A, Albert JD, et al. Shock and tissue injury induced by recombinant human cachectin. Science (Wash DC) 1986;234:470–474. - PubMed
-
- Tracey KJ, Fong Y, Hesse DG, Manogue KR, Lee AT, Kuo GC, Lowry SF, Cerami A. Anticachectin/TNF monoclonal antibodies prevent septic shock during lethal bacteraemia. Nature (Lond) 1987;330:662–664. - PubMed
-
- Tracey, K.J. 1997. Tumor necrosis factor. In Cytokines in Health and Disease. D.G. Remick and J.S. Friedland, editors. Marcel Dekker, Inc., New York. In press.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

