Axial rotation of sliding actin filaments revealed by single-fluorophore imaging
- PMID: 9159126
- PMCID: PMC20832
- DOI: 10.1073/pnas.94.11.5646
Axial rotation of sliding actin filaments revealed by single-fluorophore imaging
Abstract
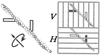
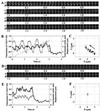
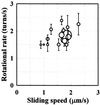
In the actomyosin motor, myosin slides along an actin filament that has a helical structure with a pitch of approximately 72 nm. Whether myosin precisely follows this helical track is an unanswered question bearing directly on the motor mechanism. Here, axial rotation of actin filaments sliding over myosin molecules fixed on a glass surface was visualized through fluorescence polarization imaging of individual tetramethylrhodamine fluorophores sparsely bound to the filaments. The filaments underwent one revolution per sliding distance of approximately 1 microm, which is much greater than the 72 nm pitch. Thus, myosin does not "walk" on the helical array of actin protomers; rather it "runs," skipping many protomers. Possible mechanisms involving sequential interaction of myosin with successive actin protomers are ruled out at least for the preparation described here in which the actin filaments ran rather slowly compared with other in vitro systems. The result also indicates that each "kick" of myosin is primarily along the axis of the actin filament. The successful, real-time observation of the changes in the orientation of a single fluorophore opens the possibility of detecting a conformational change(s) of a single protein molecule at the moment it functions.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

