Local release of GABAergic inhibition in the motor cortex induces immediate-early gene expression in indirect pathway neurons of the striatum
- PMID: 9169535
- PMCID: PMC6573341
- DOI: 10.1523/JNEUROSCI.17-12-04752.1997
Local release of GABAergic inhibition in the motor cortex induces immediate-early gene expression in indirect pathway neurons of the striatum
Abstract
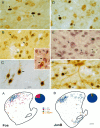
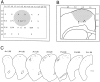
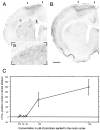
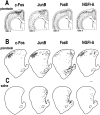
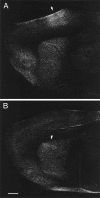
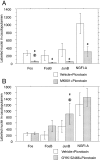
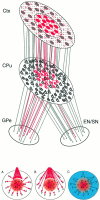
The neocortex is thought to exert a powerful influence over the functions of the basal ganglia via its projection to the striatum. It is not known, however, whether corticostriatal effects are similar across different types of striatal projection neurons and interneurons or are unique for cells having different functions within striatal networks. To examine this question, we developed a method for focal synchronous activation of the primary motor cortex (MI) of freely moving rats by local release of GABAergic inhibition. With this method, we monitored cortically evoked activation of two immediate-early gene protein products, c-Fos and JunB, in phenotypically identified striatal neurons. We further studied the influence of glutamate receptor antagonists on the stimulated expression of c-Fos, JunB, FosB, and NGFI-A. Local disinhibition of MI elicited remarkably selective induction of c-Fos and JunB in enkephalinergic projection neurons. These indirect pathway neurons, through their projections to the globus pallidus, can inhibit thalamocortical motor circuits. The dynorphin-containing projection neurons of the direct pathway, with opposite effects on the thalamocortical circuits, showed very little induction of c-Fos or JunB. The gene response of striatal interneurons was also highly selective, affecting principally parvalbumin- and NADPH diaphorase-expressing interneurons. The glutamate NMDA receptor antagonist MK-801 strongly reduced the cortically evoked striatal gene expression in all cell types for each gene examined. Because the gene induction that we found followed known corticostriatal somatotopy, was dose-dependent, and was selectively sensitive to glutamate receptor antagonists, we suggest that the differential activation patterns reflect functional specialization of cortical inputs to the direct and indirect pathways of the basal ganglia and functional plasticity within these circuits.
Figures
References
-
- Albin RL, Young AB, Penney JB. The functional anatomy of basal ganglia disorders. Trends Neurosci. 1989;12:366–375. - PubMed
-
- Albin RL, Makowiec RL, Hollingsworth ZR, Dure LS, IV, Penney JB, Young AB. Excitatory amino acid binding sites in the basal ganglia of the rat: a quantitative autoradiographic study. Neuroscience. 1992;46:35–48. - PubMed
-
- Alexander GE, Crutcher MD. Functional architecture of basal ganglia circuits: neural substrates of parallel processing. Trends Neurosci. 1990;13:266–272. - PubMed
-
- Aosaki T, Kimura M, Graybiel AM. Temporal and spatial characteristics of tonically active neurons of the primate’s striatum. J Neurophysiol. 1995;73:1234–11252. - PubMed
-
- Apicella P, Scarnati E, Ljungberg T, Schultz W. Neuronal activity in monkey striatum related to the expectation of predictable environmental events. J Neurophysiol. 1992;68:945–960. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
