Transmembrane/cytoplasmic domain-mediated membrane type 1-matrix metalloprotease docking to invadopodia is required for cell invasion
- PMID: 9223295
- PMCID: PMC21537
- DOI: 10.1073/pnas.94.15.7959
Transmembrane/cytoplasmic domain-mediated membrane type 1-matrix metalloprotease docking to invadopodia is required for cell invasion
Abstract
The invasion of human malignant melanoma cells into the extracellular matrix (ECM) involves the accumulation of proteases at sites of ECM degradation where activation of matrix metalloproteases (MMP) occurs. Here, we show that when membrane type 1 MMP (MT-MMP) was overexpressed in RPMI7951 human melanoma cells, the cells made contact with the ECM, activated soluble and ECM-bound MMP-2, and degraded and invaded the ECM. Further experiments demonstrated the importance of localization of the MT-MMP to invadopodia. Overexpression of MT-MMP without invadopodial localization caused activation of soluble MMP-2, but did not facilitate ECM degradation or cell invasiveness. Up-regulation of endogenous MT-MMP with concanavalin A caused activation of MMP-2. However, concanavalin A treatment prevented invadopodial localization of MT-MMP and ECM degradation. Neither a truncated MT-MMP mutant lacking transmembrane (TM) and cytoplasmic domains (DeltaTMMT-MMP), nor a chimeric MT-MMP containing the interleukin 2 receptor alpha chain (IL-2R) TM and cytoplasmic domains (DeltaTMMT-MMP/TMIL-2R) were localized to invadopodia or exhibited ECM degradation. Furthermore, a chimera of the TM/cytoplasmic domain of MT-MMP (TMMT-MMP) with tissue inhibitor of MMP 1 (TIMP-1/TMMT-MMP) directed the TIMP-1 molecule to invadopodia. Thus, the MT-MMP TM/cytoplasmic domain mediates the spatial organization of MT-MMP into invadopodia and subsequent degradation of the ECM.
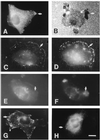
Figures



References
-
- Mignatti P, Rifkin D B. Physiol Rev. 1993;73:161–195. - PubMed
-
- Stetler-Stevenson W G, Aznavoorian S, Liotta L A. Annu Rev Cell Biol. 1993;9:541–573. - PubMed
-
- Chen W-T, Lee C C, Goldstein L, Bernier S, Liu C H, Lin C Y, Yeh Y, Monsky W L, Kelly T, Dai M, Mueller S C. Breast Cancer Res Treat. 1994;31:217–226. - PubMed
-
- Cockett M I, Birch M L, Murphy G, Hart I R, Docherty A J P. Biochem Soc Trans. 1994;22:55–57. - PubMed
-
- Birkedal-Hansen H, Moore W G I, Bodden M K, Windsor L J, Birkedal-Hansen B, DeCarlo A, Engler J A. Crit Rev Oral Biol Med. 1993;4:197–250. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

