Growth factor activity of endothelin-1 in primary astrocytes mediated by adhesion-dependent and -independent pathways
- PMID: 9236231
- PMCID: PMC6568338
- DOI: 10.1523/JNEUROSCI.17-16-06203.1997
Growth factor activity of endothelin-1 in primary astrocytes mediated by adhesion-dependent and -independent pathways
Abstract
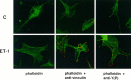
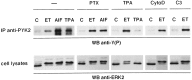
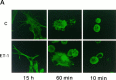
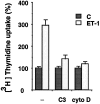
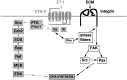
Endothelin-1 (ET-1) has been shown to induce DNA synthesis in primary astrocytes by stimulating the extracellular signal-regulated kinase (ERK) pathway. To clarify the mechanisms responsible for the anchorage-dependent growth of astrocytes, the relationships between cell adhesion and ERK activation were investigated. Here it is reported that ET-1 promotes the formation of stress fibers and focal adhesions and the tyrosine phosphorylation of focal adhesion kinase (FAK) and paxillin, as well as Src activation and association of phosphorylated FAK with Grb2. Pretreatment of astrocytes with cytochalasin D or C3-transferase, which inhibits actin polymerization or Rho activity, respectively, prevented the activation/phosphorylation of Src, FAK, and paxillin after ET-1 stimulation; by contrast, the ERK pathway was not significantly affected. This differential activation of FAK/Src and ERK pathways was also observed with astrocytes 10 and 60 min after replating on poly-L-ornithine-precoated dishes. Collectively, these findings indicate that activation of FAK and Src is dependent on actin cytoskeleton integrity, Rho activation, and adhesion to extracellular matrix, whereas ERK activation is independent of these intracellular events and seems to correlate with activation of the newly identified protein tyrosine kinase PYK2. Induction of DNA synthesis by ET-1, however, was reduced dramatically in astrocytes pretreated with either cytochalasin D or C3-transferase. This study provides a demonstration of Rho- and adhesion-dependent activation of FAK/Src, which collaborates with adhesion-independent activation of PYK2/ERK for DNA synthesis in ET-1-stimulated astrocytes.
Figures




References
-
- Cazaubon SM, Couraud PO. Nitric oxide and endothelin at the blood-brain barrier. An introduction to the blood-brain barrier Pardridge WM. 1997. Cambridge UP; Cambridge, in press.
-
- Cazaubon SM, Ramos-Morales F, Fischer S, Schweighoffer F, Strosberg AD, Couraud PO. Endothelin induces tyrosine phosphorylation and GRB2 association of SHC in astrocytes. J Biol Chem. 1994;269:24805–24809. - PubMed
-
- Couraud PO, Durieu-Trautmann O, Le Nguyen P, Marin P, Glibert F, Strosberg AD. Functional endothelin-1 receptors in rat astrocytoma C6. Eur J Pharmacol. 1991;206:191–198. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
