P2 receptor excitation of rodent hypoglossal motoneuron activity in vitro and in vivo: a molecular physiological analysis
- PMID: 9236242
- PMCID: PMC6568344
- DOI: 10.1523/JNEUROSCI.17-16-06325.1997
P2 receptor excitation of rodent hypoglossal motoneuron activity in vitro and in vivo: a molecular physiological analysis
Abstract
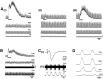
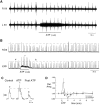
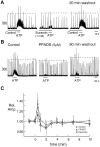
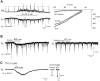
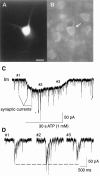
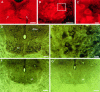
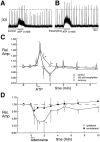
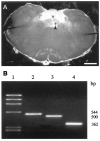
The role of P2 receptors in controlling hypoglossal motoneuron (XII MN) output was examined (1) electrophysiologically, via application of ATP to the hypoglossal nucleus of rhythmically active mouse medullary slices and anesthetized adult rats; (2) immunohistochemically, using an antiserum against the P2X2 receptor subunit; and (3) using PCR to identify expression of P2X2 receptor subunits in micropunches of tissue taken from the XII motor nucleus. Application of ATP to the hypoglossal nucleus of mouse medullary slices and anesthetized rats produced a suramin-sensitive excitation of hypoglossal nerve activity. Additional in vitro effects included potentiation of inspiratory hypoglossal nerve output via a suramin- and pyridoxal-phosphate-6-azophenyl-2',4'-disulphonic acid (PPADS)-sensitive mechanism, XII MN depolarization via activation of a suramin-sensitive inward current, decreased neuronal input resistance, and a slow-onset theophylline-sensitive reduction of inspiratory output likely resulting from hydrolysis of extracellular ATP to adenosine and activation of P1 receptors. Immunohistochemically, P2X2 receptors were detected in inspiratory XII MNs that were labeled with Lucifer yellow. These data, combined with identification of mRNA for three P2X2 receptor subunit isoforms within the hypoglossal nucleus (two of which have not been localized previously in brain) and the previous demonstration that P2X receptors are ubiquitously expressed in cranial and spinal motoneuron pools, support not only a role of P2 receptors in modulating inspiratory hypoglossal activity but a general role of P2 receptors in modulating motor outflow from the CNS.
Figures
Similar articles
-
Modulation of phrenic motoneuron excitability by ATP: consequences for respiratory-related output in vitro.J Appl Physiol (1985). 2002 May;92(5):1899-910. doi: 10.1152/japplphysiol.00475.2001. J Appl Physiol (1985). 2002. PMID: 11960940
-
Characterization of cultured dorsal root ganglion neuron P2X receptors.Eur J Neurosci. 1999 Jan;11(1):149-54. doi: 10.1046/j.1460-9568.1999.00426.x. Eur J Neurosci. 1999. PMID: 9987019
-
The use of antagonists to characterize the receptors mediating depolarization of the rat isolated vagus nerve by alpha, beta-methylene adenosine 5'-triphosphate.Br J Pharmacol. 1994 May;112(1):282-8. doi: 10.1111/j.1476-5381.1994.tb13065.x. Br J Pharmacol. 1994. PMID: 8032652 Free PMC article.
-
Modulation of hypoglossal motoneuron excitability by NK1 receptor activation in neonatal mice in vitro.J Physiol. 2001 Jul 15;534(Pt. 2):447-64. doi: 10.1111/j.1469-7793.2001.00447.x. J Physiol. 2001. PMID: 11454963 Free PMC article.
-
Synaptic control of motoneuron excitability in rodents: from months to milliseconds.Clin Exp Pharmacol Physiol. 2000 Jan-Feb;27(1-2):120-5. doi: 10.1046/j.1440-1681.2000.03202.x. Clin Exp Pharmacol Physiol. 2000. PMID: 10696540 Review.
Cited by
-
Pathophysiology of sleep apnea.Physiol Rev. 2010 Jan;90(1):47-112. doi: 10.1152/physrev.00043.2008. Physiol Rev. 2010. PMID: 20086074 Free PMC article. Review.
-
P2Y1 receptor modulation of the pre-Bötzinger complex inspiratory rhythm generating network in vitro.J Neurosci. 2007 Jan 31;27(5):993-1005. doi: 10.1523/JNEUROSCI.3948-06.2007. J Neurosci. 2007. PMID: 17267553 Free PMC article.
-
The retrotrapezoid nucleus and the neuromodulation of breathing.J Neurophysiol. 2021 Mar 1;125(3):699-719. doi: 10.1152/jn.00497.2020. Epub 2020 Dec 2. J Neurophysiol. 2021. PMID: 33427575 Free PMC article. Review.
-
Glial-derived adenosine modulates spinal motor networks in mice.J Neurophysiol. 2012 Apr;107(7):1925-34. doi: 10.1152/jn.00513.2011. Epub 2011 Dec 28. J Neurophysiol. 2012. PMID: 22205649 Free PMC article.
-
Capsaicin Enhances Glutamatergic Synaptic Transmission to Neonatal Rat Hypoglossal Motor Neurons via a TRPV1-Independent Mechanism.Front Cell Neurosci. 2017 Dec 5;11:383. doi: 10.3389/fncel.2017.00383. eCollection 2017. Front Cell Neurosci. 2017. PMID: 29259542 Free PMC article.
References
-
- Abbracchio MP, Burnstock G. Purinoceptors: are there families of P2X and P2Y purinoceptors? Pharmacol Ther. 1994;64:445–475. - PubMed
-
- Aldes LD, Chapman ME, Chronister RB, Haycock JW. Sources of noradrenergic afferents to the hypoglossal nucleus of the rat. Brain Res Bull. 1992;29:931–942. - PubMed
-
- Akasu T, Hirai K, Koketsu K. Modulatory actions of ATP on membrane potentials of bullfrog sympathetic ganglion cells. Brain Res. 1983;258:313–317. - PubMed
-
- Bellingham MC, Berger AJ. Adenosine suppresses excitatory glutamatergic inputs to rat hypoglossal motoneurons in vitro. Neurosci Lett. 1994;177:143–146. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
