Physiology and plasticity of morphologically identified cells in the mormyrid electrosensory lobe
- PMID: 9236249
- PMCID: PMC6568337
- DOI: 10.1523/JNEUROSCI.17-16-06409.1997
Physiology and plasticity of morphologically identified cells in the mormyrid electrosensory lobe
Abstract
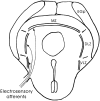
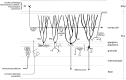
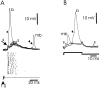
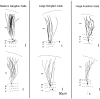
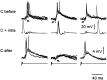
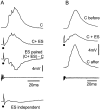
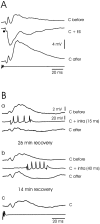
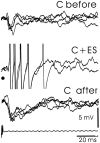
The electrosensory lobe (ELL) of mormyrid electric fish is the first stage in the central processing of sensory input from electroreceptors. The responses of cells in ELL to electrosensory input are strongly affected by corollary discharge signals associated with the motor command that drives the electric organ discharge (EOD). This study used intracellular recording and staining to describe the physiology of three major cell types in the mormyrid ELL: the medium ganglion cell, the large ganglion cell, and the large fusiform cell. The medium ganglion cell is a Purkinje-like interneuron, whereas the large ganglion and large fusiform cells are efferent neurons that convey electrosensory information to higher stages of the system. Clear differences were observed among the three cell types. Medium ganglion cells showed two types of spikes, a small narrow spike and a large broad spike that were probably of axonal and dendro-somatic origin, respectively, whereas the large ganglion and large fusiform cells showed only large narrow spikes. Most of the medium ganglion cells and all of the large ganglion cells were inhibited by electrosensory stimuli in the center of their receptive fields, whereas the large fusiform cells were excited by such stimuli. Responses to the EOD corollary discharge were different in the three cell types, and these responses underwent plastic changes after a few minutes of pairing with an electrosensory stimulus. Plastic changes were also observed in medium and large ganglion cells after the corollary discharge was paired with depolarizing, intracellular current pulses.
Figures




Similar articles
-
The mormyromast region of the mormyrid electrosensory lobe. I. Responses to corollary discharge and electrosensory stimuli.J Neurophysiol. 2003 Aug;90(2):1193-210. doi: 10.1152/jn.00211.2003. J Neurophysiol. 2003. PMID: 12904505
-
Sensory processing and corollary discharge effects in mormyromast regions of mormyrid electrosensory lobe. II. Cell types and corollary discharge plasticity.J Neurophysiol. 1992 Sep;68(3):859-75. doi: 10.1152/jn.1992.68.3.859. J Neurophysiol. 1992. PMID: 1432053
-
Physiology of electrosensory lateral line lobe neurons in Gnathonemus petersii.J Exp Biol. 1999 May;202(Pt 10):1301-9. doi: 10.1242/jeb.202.10.1301. J Exp Biol. 1999. PMID: 10210670 Review.
-
Nucleus preeminentialis of mormyrid fish, a center for recurrent electrosensory feedback. I. Electrosensory and corollary discharge responses.J Neurophysiol. 1996 Sep;76(3):1581-96. doi: 10.1152/jn.1996.76.3.1581. J Neurophysiol. 1996. PMID: 8890278
-
The role of motor command feedback in electrosensory processing.Eur J Morphol. 1994 Aug;32(2-4):225-34. Eur J Morphol. 1994. PMID: 7803171 Review.
Cited by
-
Transformations of electrosensory encoding associated with an adaptive filter.J Neurosci. 2008 Feb 13;28(7):1598-612. doi: 10.1523/JNEUROSCI.4946-07.2008. J Neurosci. 2008. PMID: 18272681 Free PMC article.
-
Plastic corollary discharge predicts sensory consequences of movements in a cerebellum-like circuit.Neuron. 2014 May 21;82(4):896-907. doi: 10.1016/j.neuron.2014.03.025. Neuron. 2014. PMID: 24853945 Free PMC article.
-
Effects of sensing behavior on a latency code.J Neurosci. 2006 Aug 9;26(32):8221-34. doi: 10.1523/JNEUROSCI.1508-06.2006. J Neurosci. 2006. PMID: 16899717 Free PMC article.
-
Anti-hebbian spike-timing-dependent plasticity and adaptive sensory processing.Front Comput Neurosci. 2010 Dec 31;4:156. doi: 10.3389/fncom.2010.00156. eCollection 2010. Front Comput Neurosci. 2010. PMID: 21228915 Free PMC article.
-
Physiological evidence of sensory integration in the electrosensory lateral line lobe of Gnathonemus petersii.PLoS One. 2018 Apr 11;13(4):e0194347. doi: 10.1371/journal.pone.0194347. eCollection 2018. PLoS One. 2018. PMID: 29641541 Free PMC article.
References
-
- Bastian J. Pyramidal-cell plasticity in weakly electric fish: a mechanism for attenuating responses to reafferent electrosensory inputs. J Comp Physiol. 1995;176:63–78. - PubMed
-
- Bastian J. Plasticity in an electrosensory system. II. Postsynaptic events associated with a dynamic sensory filter. J Neurophysiol. 1996;76:2497–2507. - PubMed
-
- Bell CC. An efference copy modified by reafferent input. Science. 1981;214:450–453. - PubMed
-
- Bell CC. Properties of a modifiable efference copy in electric fish. J Neurophysiol. 1982;47:1043–1056. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
