Antibody-mediated inhibition of the growth of larvae from an insect causing cutaneous myiasis in a mammalian host
- PMID: 9256413
- PMCID: PMC22971
- DOI: 10.1073/pnas.94.17.8939
Antibody-mediated inhibition of the growth of larvae from an insect causing cutaneous myiasis in a mammalian host
Abstract
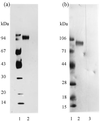
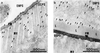
Many insects feed on blood or tissue from mammalian hosts. One potential strategy for the control of these insects is to vaccinate the host with antigens derived from the insect. The larvae of the fly Lucilia cuprina feed on ovine tissue and tissue fluids causing a cutaneous myiasis associated with considerable host morbidity and mortality. A candidate vaccine antigen, peritrophin 95, was purified from the peritrophic membrane, which lines the gut of these larvae. Serum from sheep vaccinated with peritrophin 95 inhibited growth of first-instar L. cuprina larvae that fed on this serum. Growth inhibition was probably caused by antibody-mediated blockage of the normally semipermeable peritrophic membrane and the subsequent development of an impervious layer of undefined composition on the gut lumen side of the peritrophic membrane that restricted access of nutrients to the larvae. The amino acid sequence of peritrophin 95 was determined by cloning the DNA complementary to its mRNA. The deduced amino acid sequence codes for a secreted protein containing a distinct Cys-rich domain of 317 amino acids followed by a mucin-like domain of 139 amino acids. The Cys-rich domain may be involved in binding chitin. This report describes a novel immunological strategy for the potential control of L. cuprina larvae that may have general application to the control of other insect pests.
Figures



Similar articles
-
Characterization of a major peritrophic membrane protein, peritrophin-44, from the larvae of Lucilia cuprina. cDNA and deduced amino acid sequences.J Biol Chem. 1996 Apr 12;271(15):8925-35. doi: 10.1074/jbc.271.15.8925. J Biol Chem. 1996. PMID: 8621536
-
Role of oligosaccharides in the immune response of sheep vaccinated with Lucilia cuprina larval glycoprotein, peritrophin-95.Int J Parasitol. 2001 Jun;31(8):798-809. doi: 10.1016/s0020-7519(01)00195-3. Int J Parasitol. 2001. PMID: 11403771 Clinical Trial.
-
cDNA and deduced amino acid sequences of a peritrophic membrane glycoprotein, 'peritrophin-48', from the larvae of Lucilia cuprina.Insect Biochem Mol Biol. 1998 Feb;28(2):99-111. doi: 10.1016/s0965-1748(97)00103-3. Insect Biochem Mol Biol. 1998. PMID: 9639876
-
Vaccination against Lucilia cuprina: the causative agent of sheep blowfly strike.Immunol Cell Biol. 1993 Oct;71 ( Pt 5):453-62. doi: 10.1038/icb.1993.51. Immunol Cell Biol. 1993. PMID: 8270274 Review.
-
Cutaneous myiasis. Recent advances in biology, immunology and improvements of the control measures.Ann Parasitol Hum Comp. 1991;66 Suppl 1:52-7. Ann Parasitol Hum Comp. 1991. PMID: 1805677 Review.
Cited by
-
Identification and Characterization of a 25 kDa Protein That Is Indispensable for the Efficient Saccharification of Eisenia bicyclis in the Digestive Fluid of Aplysia kurodai.PLoS One. 2017 Jan 27;12(1):e0170669. doi: 10.1371/journal.pone.0170669. eCollection 2017. PLoS One. 2017. PMID: 28129373 Free PMC article.
-
Identification and molecular characterization of a chitin-binding protein from the beet webworm, Loxostege sticticalis L.Int J Mol Sci. 2014 Oct 22;15(10):19147-61. doi: 10.3390/ijms151019147. Int J Mol Sci. 2014. PMID: 25340980 Free PMC article.
-
Identification of a polymorphic mucin-like gene expressed in the midgut of the mosquito, Aedes aegypti, using an integrated bulked segregant and differential display analysis.Genetics. 2001 Jul;158(3):1125-36. doi: 10.1093/genetics/158.3.1125. Genetics. 2001. PMID: 11454761 Free PMC article.
-
Molecular characterization of a peritrophic membrane protein from the silkworm, Bombyx mori.Mol Biol Rep. 2013 Feb;40(2):1087-95. doi: 10.1007/s11033-012-2151-5. Epub 2012 Oct 14. Mol Biol Rep. 2013. PMID: 23065286
-
Lucilia cuprina genome unlocks parasitic fly biology to underpin future interventions.Nat Commun. 2015 Jun 25;6:7344. doi: 10.1038/ncomms8344. Nat Commun. 2015. PMID: 26108605 Free PMC article.
References
-
- Tellam R L, Smith D, Kemp D H, Willadsen P. In: Animal Parasite Control Using Biotechnology. Wong W K, editor. London: CRC; 1992. pp. 303–331.
-
- Willadsen P, Eisemann C H, Tellam R L. Parasitol Today. 1993;9:132–135. - PubMed
-
- Tellam R L. In: The Biology of the Insect Midgut. Lehane M J, Billingsley P F, editors. London: Chapman & Hall; 1996. pp. 87–113.
-
- Eisemann C H, Pearson R D, Donaldson R A, Cadogan L C, Vuocolo T. Med Vet Entomol. 1993;7:177–185. - PubMed
-
- East I J, Fitzgerald C J, Pearson R D, Donaldson R A, Vuocolo T, Cadogan L C, Eisemann C H, Tellam R L. Int J Parasitol. 1993;23:221–229. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

