Eosinophil lipid bodies: specific, inducible intracellular sites for enhanced eicosanoid formation
- PMID: 9294145
- PMCID: PMC2199047
- DOI: 10.1084/jem.186.6.909
Eosinophil lipid bodies: specific, inducible intracellular sites for enhanced eicosanoid formation
Abstract
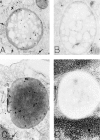
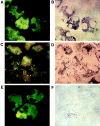
The specific intracellular sites at which enzymes act to generate arachidonate-derived eicosanoid mediators of inflammation are uncertain. We evaluated the formation and function of cytoplasmic lipid bodies. Lipid body formation in eosinophils was a rapidly (<1 h) inducible response which was platelet-activating factor (PAF) receptor-mediated, involved signaling through protein kinase C, and required new protein synthesis. In intact and enucleated eosinophils, the PAF-induced increases in lipid body numbers correlated with enhanced production of both lipoxygenase- and cyclooxygenase-derived eicosanoids. All principal eosinophil eicosanoid-forming enzymes, 5-lipoxygenase, leukotriene C4 synthase, and cyclooxygenase, were immunolocalized to native as well as newly induced lipid bodies in intact and enucleated eosinophils. Thus, lipid bodies are structurally distinct, inducible, nonnuclear sites for enhanced synthesis of paracrine eicosanoid mediators of inflammation.
Figures
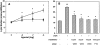



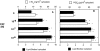

Similar articles
-
Lipopolysaccharide-induced leukocyte lipid body formation in vivo: innate immunity elicited intracellular Loci involved in eicosanoid metabolism.J Immunol. 2002 Dec 1;169(11):6498-506. doi: 10.4049/jimmunol.169.11.6498. J Immunol. 2002. PMID: 12444160
-
Mechanisms of platelet-activating factor-induced lipid body formation: requisite roles for 5-lipoxygenase and de novo protein synthesis in the compartmentalization of neutrophil lipids.J Exp Med. 1996 Apr 1;183(4):1515-25. doi: 10.1084/jem.183.4.1515. J Exp Med. 1996. PMID: 8666909 Free PMC article.
-
Pathways for eosinophil lipid body induction: differing signal transduction in cells from normal and hypereosinophilic subjects.J Leukoc Biol. 1998 Oct;64(4):563-9. doi: 10.1002/jlb.64.4.563. J Leukoc Biol. 1998. PMID: 9766638
-
The cellular biology of eosinophil eicosanoid formation and function.J Allergy Clin Immunol. 2002 Mar;109(3):393-400. doi: 10.1067/mai.2002.121529. J Allergy Clin Immunol. 2002. PMID: 11897981 Review.
-
Mechanisms of formation and function of eosinophil lipid bodies: inducible intracellular sites involved in arachidonic acid metabolism.Mem Inst Oswaldo Cruz. 1997;92 Suppl 2:135-40. doi: 10.1590/s0074-02761997000800018. Mem Inst Oswaldo Cruz. 1997. PMID: 9698925 Review.
Cited by
-
Adipose triglyceride lipase regulates eicosanoid production in activated human mast cells.J Lipid Res. 2014 Dec;55(12):2471-8. doi: 10.1194/jlr.M048553. Epub 2014 Aug 11. J Lipid Res. 2014. PMID: 25114172 Free PMC article.
-
A mechanism of benefit of soy genistein in asthma: inhibition of eosinophil p38-dependent leukotriene synthesis.Clin Exp Allergy. 2008 Jan;38(1):103-12. doi: 10.1111/j.1365-2222.2007.02862.x. Epub 2007 Nov 2. Clin Exp Allergy. 2008. PMID: 17979994 Free PMC article. Clinical Trial.
-
Leukocyte lipid bodies - Biogenesis and functions in inflammation.Biochim Biophys Acta. 2009 Jun;1791(6):540-51. doi: 10.1016/j.bbalip.2009.01.005. Epub 2009 Jan 21. Biochim Biophys Acta. 2009. PMID: 19416659 Free PMC article. Review.
-
Allergic challenge-elicited lipid bodies compartmentalize in vivo leukotriene C4 synthesis within eosinophils.Am J Respir Cell Mol Biol. 2005 Sep;33(3):254-61. doi: 10.1165/rcmb.2005-0145OC. Epub 2005 Jun 9. Am J Respir Cell Mol Biol. 2005. PMID: 15947420 Free PMC article.
-
Enhancement of lipid bodies during differentiation of skeletal myofibroblasts of rat's fetus in vitro.In Vitro Cell Dev Biol Anim. 2004 Jan-Feb;40(1-2):1-3. doi: 10.1290/1543-706X(2004)40<1:EOLBDD>2.0.CO;2. In Vitro Cell Dev Biol Anim. 2004. PMID: 15180437
References
-
- Serhan CN. Eicosanoids in leukocyte function. Curr Opin Hematol. 1994;1:69–77. - PubMed
-
- Henderson WR., Jr the role of leukotrienes in inflammation. Ann Intern Med. 1994;121:684–697. - PubMed
-
- Morita I, Schindler M, Regier MK, Otto JC, Hori T, DeWitt DL, Smith WL. Different intracellular locations for prostaglandin endoperoxide H synthase-1 and -2. J Biol Chem. 1995;270:10902–10908. - PubMed
-
- Otto JC, Smith WL. The orientation of prostaglandin endoperoxide synthases-1 and -2 in the endoplasmic reticulum. J Biol Chem. 1994;269:19868–19875. - PubMed
-
- Smith WL, Garavito RM, DeWitt DL. Prostaglandin endoperoxide H synthases (cyclooxygenases)-1 and -2. J Biol Chem. 1996;271:33157–33160. - PubMed

