Antagonism of cell adhesion by an alpha-catenin mutant, and of the Wnt-signaling pathway by alpha-catenin in Xenopus embryos
- PMID: 9362521
- PMCID: PMC2139960
- DOI: 10.1083/jcb.139.4.1033
Antagonism of cell adhesion by an alpha-catenin mutant, and of the Wnt-signaling pathway by alpha-catenin in Xenopus embryos
Abstract
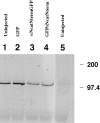
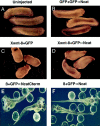
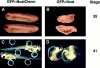
In Xenopus laevis development, beta-catenin plays an important role in the Wnt-signaling pathway by establishing the Nieuwkoop center, which in turn leads to specification of the dorsoventral axis. Cadherins are essential for embryonic morphogenesis since they mediate calcium-dependent cell-cell adhesion and can modulate beta-catenin signaling. alpha-catenin links beta-catenin to the actin-based cytoskeleton. To study the role of endogenous alpha-catenin in early development, we have made deletion mutants of alphaN-catenin. The binding domain of beta-catenin has been mapped to the NH2-terminal 210 amino acids of alphaN-catenin. Overexpression of mutants lacking the COOH-terminal 230 amino acids causes severe developmental defects that reflect impaired calcium-dependent blastomere adhesion. Lack of normal adhesive interactions results in a loss of the blastocoel in early embryos and ripping of the ectodermal layer during gastrulation. The phenotypes of the dominant-negative mutants can be rescued by coexpressing full-length alphaN-catenin or a mutant of beta-catenin that lacks the internal armadillo repeats. We next show that coexpression of alphaN-catenin antagonizes the dorsalizing effects of beta-catenin and Xwnt-8. This can be seen phenotypically, or by studying the effects of expression on the downstream homeobox gene Siamois. Thus, alpha-catenin is essential for proper morphogenesis of the embryo and may act as a regulator of the intracellular beta-catenin signaling pathway in vivo.
Figures










References
-
- Aberle H, Schwartz H, Hoschuetzky H, Kemler R. Single amino acid substitutions in proteins of the armadillo gene family abolish their binding to α-catenin. J Biol Chem. 1996;271:1520–1526. - PubMed
-
- Behrens J, von Kries JP, Kühl M, Bruhn L, Wedlich D, Grosschedl R, Birchmeier W. Functional interaction of β-catenin with the transcription factor Lef-1. Nature. 1996;382:638–642. - PubMed
-
- Brannon M, Kimelman D. Activation of Siamoisby the Wnt pathway. Dev Biol. 1996;180:344–347. - PubMed

