The Aer protein and the serine chemoreceptor Tsr independently sense intracellular energy levels and transduce oxygen, redox, and energy signals for Escherichia coli behavior
- PMID: 9380671
- PMCID: PMC23396
- DOI: 10.1073/pnas.94.20.10541
The Aer protein and the serine chemoreceptor Tsr independently sense intracellular energy levels and transduce oxygen, redox, and energy signals for Escherichia coli behavior
Abstract
We identified a protein, Aer, as a signal transducer that senses intracellular energy levels rather than the external environment and that transduces signals for aerotaxis (taxis to oxygen) and other energy-dependent behavioral responses in Escherichia coli. Domains in Aer are similar to the signaling domain in chemotaxis receptors and the putative oxygen-sensing domain of some transcriptional activators. A putative FAD-binding site in the N-terminal domain of Aer shares a consensus sequence with the NifL, Bat, and Wc-1 signal-transducing proteins that regulate gene expression in response to redox changes, oxygen, and blue light, respectively. A double mutant deficient in aer and tsr, which codes for the serine chemoreceptor, was negative for aerotaxis, redox taxis, and glycerol taxis, each of which requires the proton motive force and/or electron transport system for signaling. We propose that Aer and Tsr sense the proton motive force or cellular redox state and thereby integrate diverse signals that guide E. coli to environments where maximal energy is available for growth.
Figures

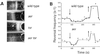
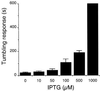
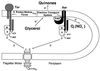
References
-
- Taylor B L. Trends Biochem Sci. 1983;8:438–441.
-
- Taylor B L. Annu Rev Microbiol. 1983;37:551–573. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

