Regulation of the receptor specificity and function of the chemokine RANTES (regulated on activation, normal T cell expressed and secreted) by dipeptidyl peptidase IV (CD26)-mediated cleavage
- PMID: 9382885
- PMCID: PMC2199148
- DOI: 10.1084/jem.186.11.1865
Regulation of the receptor specificity and function of the chemokine RANTES (regulated on activation, normal T cell expressed and secreted) by dipeptidyl peptidase IV (CD26)-mediated cleavage
Abstract
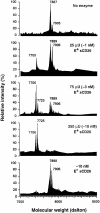
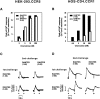
CD26 is a leukocyte activation marker that possesses dipeptidyl peptidase IV activity but whose natural substrates and immunological functions have not been clearly defined. Several chemo-kines, including RANTES (regulated on activation, normal T cell expressed and secreted), have now been shown to be substrates for recombinant soluble human CD26. The truncated RANTES(3-68) lacked the ability of native RANTES(1-68) to increase the cytosolic calcium concentration in human monocytes, but still induced this response in macrophages activated with macrophage colony-stimulating factor. Analysis of chemokine receptor messenger RNAs and patterns of desensitization of chemokine responses showed that the differential activity of the truncated molecule results from an altered receptor specificity. RANTES(3-68) showed a reduced activity, relative to that of RANTES(1-68), with cells expressing the recombinant CCR1 chemokine receptor, but retained the ability to stimulate CCR5 receptors and to inhibit the cytopathic effects of HIV-1. Our results indicate that CD26-mediated processing together with cell activation-induced changes in receptor expression provides an integrated mechanism for differential cell recruitment and for the regulation of target cell specificity of RANTES, and possibly other chemokines.
Figures





References
-
- Murphy PM. Chemokine receptors: structure, function and role in microbial pathogenesis. Cytokine Growth Factor Rev. 1996;7:47–64. - PubMed
-
- Gong J-H, Uguccioni M, Dewald B, Baggiolini M, Clark-Lewis I. RANTES and MCP-3 antagonists bind multiple chemokine receptors. J Biol Chem. 1996;271:10521–10527. - PubMed
-
- Arenzana-Seisdedos F, Virelizier J-L, Rousset D, Clark-Lewis I, Loetscher P, Moser B, Baggiolini M. HIV blocked by chemokine antagonist. Nature. 1996;383:400. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

