Nerve growth factor modulates synaptic transmission between sympathetic neurons and cardiac myocytes
- PMID: 9391012
- PMCID: PMC6573427
- DOI: 10.1523/JNEUROSCI.17-24-09573.1997
Nerve growth factor modulates synaptic transmission between sympathetic neurons and cardiac myocytes
Abstract
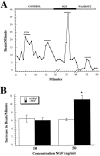
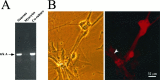
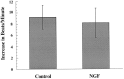
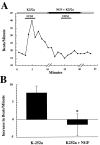
Regulation of heart rate by the sympathetic nervous system involves the release of norepinephrine (NE) from nerve terminals onto heart tissue, resulting in an elevation in beat rate. Nerve growth factor (NGF) is a neurotrophin produced by the heart that supports the survival and differentiation of sympathetic neurons. Here we report that NGF also functions as a modulator of sympathetic synaptic transmission. We determined the effect of NGF on the strength of synaptic transmission in co-cultures of neonatal rat cardiac myocytes and sympathetic neurons from the superior cervical ganglion (SCG). Synaptic transmission was assayed functionally, as an increase in the beat rate of a cardiac myocyte during stimulation of a connected neuron. Application of NGF produced a pronounced, reversible enhancement of synaptic strength. We found that TrkA, the receptor tyrosine kinase that mediates many NGF responses, is expressed primarily by neurons in these cultures, suggesting a presynaptic mechanism for the effects of NGF. A presynaptic model is further supported by the finding that NGF did not alter the response of myocytes to application of NE. In addition to the acute modulatory effects of NGF, we found that the concentration of NGF in the growth medium affects the level of synaptic transmission in cultures of sympathetic neurons and cardiac myocytes. These results indicate that in addition to its role as a survival factor, NGF plays both acute and long-term roles in the regulation of developing sympathetic synapses in the cardiac system.
Figures



References
-
- Armour JA, Ardell JL. Neurocardiology. Oxford UP; New York: 1994.
-
- Birren SJ, Lo LC, Anderson DJ. Sympathetic neurons undergo a developmental switch in trophic dependence. Development. 1993;119:597–610. - PubMed
-
- Buckley KM, Landis SC. Morphological studies of synapses and varicosities in dissociated cell cultures of sympathetic neurons. J Neurocytol. 1983;12:67–92. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
