Impaired parallel fiber-->Purkinje cell synapse stabilization during cerebellar development of mutant mice lacking the glutamate receptor delta2 subunit
- PMID: 9391016
- PMCID: PMC6573399
- DOI: 10.1523/JNEUROSCI.17-24-09613.1997
Impaired parallel fiber-->Purkinje cell synapse stabilization during cerebellar development of mutant mice lacking the glutamate receptor delta2 subunit
Abstract
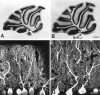
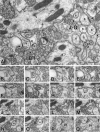
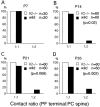
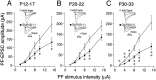
The glutamate receptor delta2 subunit (GluRdelta2) is specifically expressed in cerebellar Purkinje cells (PCs) from early developmental stages and is selectively localized at dendritic spines forming synapses with parallel fibers (PFs). Targeted disruption of the GluRdelta2 gene leads to a significant reduction of PF-->PC synapses. To address its role in the synaptogenesis, the morphology and electrophysiology of PF-->PC synapses were comparatively examined in developing GluRdelta2 mutant and wild-type cerebella. PCs in GluRdelta2 mutant mice were normally produced, migrated, and formed spines, as did those in wild-type mice. At the end of the first postnatal week, 74-78% of PC spines in both mice formed immature synapses, which were characterized by small synaptic contact, few synaptic vesicles, and incomplete surrounding by astroglial processes, eliciting little electrophysiological response. During the second and third postnatal weeks when spines and terminals are actively generated, the percentage of PC spines forming synapses attained 98-99% in wild type but remained as low as 55-60% in mutants, and the rest were unattached to any nerve terminals. As a result, the number of PF synapses per single-mutant PCs was reduced to nearly a half-level of wild-type PCs. Parallelly, PF stimulation less effectively elicited EPSCs in mutant PCs than in wild-type PCs during and after the second postnatal week. These results suggest that the GluRdelta2 is involved in the stabilization and strengthening of synaptic connectivity between PFs and PCs, leading to the association of all PC spines with PF terminals to form functionally mature synapses.
Figures




Similar articles
-
Roles of glutamate receptor delta 2 subunit (GluRdelta 2) and metabotropic glutamate receptor subtype 1 (mGluR1) in climbing fiber synapse elimination during postnatal cerebellar development.J Neurosci. 2001 Dec 15;21(24):9701-12. doi: 10.1523/JNEUROSCI.21-24-09701.2001. J Neurosci. 2001. PMID: 11739579 Free PMC article.
-
Regulation of long-term depression and climbing fiber territory by glutamate receptor delta2 at parallel fiber synapses through its C-terminal domain in cerebellar Purkinje cells.J Neurosci. 2007 Oct 31;27(44):12096-108. doi: 10.1523/JNEUROSCI.2680-07.2007. J Neurosci. 2007. PMID: 17978051 Free PMC article.
-
Influence of parallel fiber-Purkinje cell synapse formation on postnatal development of climbing fiber-Purkinje cell synapses in the cerebellum.Neuroscience. 2009 Sep 1;162(3):601-11. doi: 10.1016/j.neuroscience.2008.12.037. Epub 2008 Dec 31. Neuroscience. 2009. PMID: 19166909 Review.
-
Glutamate receptor δ2 is essential for input pathway-dependent regulation of synaptic AMPAR contents in cerebellar Purkinje cells.J Neurosci. 2011 Mar 2;31(9):3362-74. doi: 10.1523/JNEUROSCI.5601-10.2011. J Neurosci. 2011. PMID: 21368048 Free PMC article.
-
Glutamate-receptor-like molecule GluRδ2 involved in synapse formation at parallel fiber-Purkinje neuron synapses.Cerebellum. 2012 Mar;11(1):71-7. doi: 10.1007/s12311-010-0170-0. Cerebellum. 2012. PMID: 20387025 Review.
Cited by
-
Glutamate receptor delta2 subunit in activity-dependent heterologous synaptic competition.J Neurosci. 2003 Mar 15;23(6):2363-70. doi: 10.1523/JNEUROSCI.23-06-02363.2003. J Neurosci. 2003. PMID: 12657696 Free PMC article.
-
Carbonic anhydrase related protein 8 mutation results in aberrant synaptic morphology and excitatory synaptic function in the cerebellum.Mol Cell Neurosci. 2007 May;35(1):161-70. doi: 10.1016/j.mcn.2007.02.013. Epub 2007 Feb 23. Mol Cell Neurosci. 2007. PMID: 17376701 Free PMC article.
-
Characterization of trans-neuronal trafficking of Cbln1.Mol Cell Neurosci. 2009 Jun;41(2):258-73. doi: 10.1016/j.mcn.2009.03.005. Epub 2009 Apr 1. Mol Cell Neurosci. 2009. PMID: 19344768 Free PMC article.
-
Activation of steroid-sensitive TRPM3 channels potentiates glutamatergic transmission at cerebellar Purkinje neurons from developing rats.J Neurochem. 2011 Nov;119(3):474-85. doi: 10.1111/j.1471-4159.2011.07441.x. Epub 2011 Sep 28. J Neurochem. 2011. PMID: 21955047 Free PMC article.
-
Nasal obstruction during development leads to defective synapse elimination, hypersynchrony, and impaired cerebellar function.Commun Biol. 2024 Oct 23;7(1):1381. doi: 10.1038/s42003-024-07095-4. Commun Biol. 2024. PMID: 39443666 Free PMC article.
References
-
- Altman J. Postnatal development of the cerebellar cortex in the rat. I. The external germinal layer and the transitional molecular layer. J Comp Neurol. 1972a;145:353–398. - PubMed
-
- Altman J. Postnatal development of the cerebellar cortex in the rat. II. Phases in the maturation of Purkinje cells and of the molecular layer. J Comp Neurol. 1972b;145:399–463. - PubMed
-
- Altman J. Postnatal development of the cerebellar cortex in the rat. III. Maturation of the components of the granular layer. J Comp Neurol. 1972c;145:465–514. - PubMed
-
- Altman J. Morphological development of the rat cerebellum and some of its mechanisms. Exp Brain Res [Suppl] 1982;6:8–49.
-
- Altman J, Anderson WJ. Experimental reorganization of the cerebellar cortex. I. Morphological effects of elimination of all microneurons with prolonged x-irradiation started at birth. J Comp Neurol. 1972;146:355–406. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
