Apparent mtDNA heteroplasmy in Alzheimer's disease patients and in normals due to PCR amplification of nucleus-embedded mtDNA pseudogenes
- PMID: 9405710
- PMCID: PMC25134
- DOI: 10.1073/pnas.94.26.14894
Apparent mtDNA heteroplasmy in Alzheimer's disease patients and in normals due to PCR amplification of nucleus-embedded mtDNA pseudogenes
Abstract
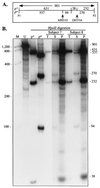
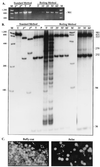
In an unprecedented finding, Davis et al. [Davis, R. E., Miller, S., Herrnstadt, C., Ghosh, S. S., Fahy, E., Shinobu, L. A., Galasko, D., Thal, L. J., Beal, M. F., Howell, N. & Parker, W. D., Jr. (1997) Proc. Natl. Acad. Sci. USA 94, 4526-4531] used an unusual DNA isolation method to show that healthy adults harbor a specific population of mutated mitochondrial cytochrome c oxidase (COX) genes that coexist with normal mtDNAs. They reported that this heteroplasmic population was present at a level of 10-15% in the blood of normal individuals and at a significantly higher level (20-30%) in patients with sporadic Alzheimer's disease. We provide compelling evidence that the DNA isolation method employed resulted in the coamplification of authentic mtDNA-encoded COX genes together with highly similar COX-like sequences embedded in nuclear DNA ("mtDNA pseudogenes"). We conclude that the observed heteroplasmy is an artifact.
Figures



References
-
- Goate A, Chartier-Harlin M-C, Mullan M, Brown J, Crawford F, et al. Nature (London) 1991;349:704–706. - PubMed
-
- Sherrington R, Rogaev E I, Liang Y, Rogaeva E A, Levesque G, et al. Nature (London) 1995;375:754–760. - PubMed
-
- Levy-Lahad E, Bird T D. Ann Neurol. 1996;40:829–840. - PubMed
-
- Levy-Lahad E, Wasco W, Poorkaj P, Romano D M, Oshima J, Pettingell W H, Yu C-E, Jondro P D, Schmidt S D, Wand K, Crowley A C, Fu Y H, Fuenette SY, Galas D, Nemens E, Wijsmann E M, Bird TD, Schellenberg GD, Tanzi R E. Science. 1995;269:973–977. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

