Sorting of beta-actin mRNA and protein to neurites and growth cones in culture
- PMID: 9412505
- PMCID: PMC6793411
- DOI: 10.1523/JNEUROSCI.18-01-00251.1998
Sorting of beta-actin mRNA and protein to neurites and growth cones in culture
Abstract
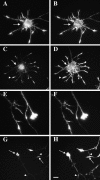
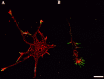
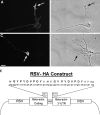
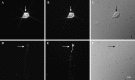
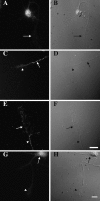
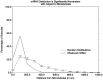
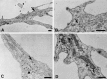
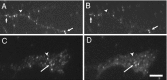
The transport of mRNAs into developing dendrites and axons may be a basic mechanism to localize cytoskeletal proteins to growth cones and influence microfilament organization. Using isoform-specific antibodies and probes for in situ hybridization, we observed distinct localization patterns for beta- and gamma-actin within cultured cerebrocortical neurons. beta-Actin protein was highly enriched within growth cones and filopodia, in contrast to gamma-actin protein, which was distributed uniformly throughout the cell. beta-Actin protein also was shown to be peripherally localized after transfection of beta-actin cDNA bearing an epitope tag. beta-Actin mRNAs were localized more frequently to neuronal processes and growth cones, unlike gamma-actin mRNAs, which were restricted to the cell body. The rapid localization of beta-actin mRNA, but not gamma-actin mRNA, into processes and growth cones could be induced by dibutyryl cAMP treatment. Using high-resolution in situ hybridization and image-processing methods, we showed that the distribution of beta-actin mRNA within growth cones was statistically nonrandom and demonstrated an association with microtubules. beta-Actin mRNAs were detected within minor neurites, axonal processes, and growth cones in the form of spatially distinct granules that colocalized with translational components. Ultrastructural analysis revealed polyribosomes within growth cones that colocalized with cytoskeletal filaments. The transport of beta-actin mRNA into developing neurites may be a sequence-specific mechanism to synthesize cytoskeletal proteins directly within processes and growth cones and would provide an additional means to deliver cytoskeletal proteins over long distances.
Figures


References
-
- Barbarese E, Koppel DE, Deutscher MP, Smith CL, Ainger K, Morgan F, Carson JH. Protein translation components are colocalized in granules in oligodendrocytes. J Cell Sci. 1995;108:2781–2790. - PubMed
-
- Bassell GJ, Singer RH. mRNA and cytoskeletal filaments. Curr Opin Cell Biol. 1997;9:109–115. - PubMed
-
- Bassell GJ, Singer RH, Kosik KS. Association of poly(A+) mRNA with microtubules in cultured neurons. Neuron. 1994;12:571–582. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
