Organization and transmitter specificity of medullary neurons activated by sustained hypertension: implications for understanding baroreceptor reflex circuitry
- PMID: 9412514
- PMCID: PMC6793387
- DOI: 10.1523/JNEUROSCI.18-01-00371.1998
Organization and transmitter specificity of medullary neurons activated by sustained hypertension: implications for understanding baroreceptor reflex circuitry
Abstract
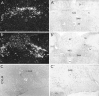
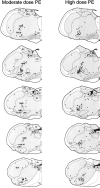
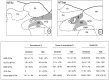
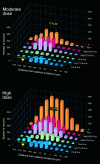
In situ expression of c-fos observed in response to phenylephrine (PE)-induced hypertension provided a basis for characterizing the organization and neurotransmitter specificity of neurons at nodal points of medullary baroreflex circuitry. Sustained hypertension induced by a moderate dose of PE provoked patterns of c-fos mRNA and protein expression that conformed in the nucleus of the solitary tract (NTS) to the termination patterns of primary baroreceptor afferents and in the caudal ventrolateral medulla (CVLM) to a physiologically defined depressor region. A majority of barosensitive CVLM neurons concurrently displayed markers for the GABAergic phenotype; few were glycinergic. Phenylephrine-sensitive GABAergic neurons that were retrogradely labeled after tracer deposits in pressor sites of the rostral ventrolateral medulla (RVLM) occupied a zone extending approximately 1.4 mm rostrally from the level of the calamus scriptorius, intermingled partly with catecholaminergic neurons of the A1 and C1 cell groups. By contrast, barosensitive neurons of the NTS were found to be phenotypically complex, with very few projecting directly to the RVLM. Extensive colocalization of PE-induced Fos-IR and markers for the nitric oxide phenotype were seen in a circumscribed, rostral, portion of the baroreceptor afferent zone of the NTS, whereas only a small proportion of PE-sensitive neurons in the NTS were found to be GABAergic. PE treatment parameters have been identified that provide a basis for defining and characterizing populations of neurons at the first station in the central processing of primary baroreceptor input and at a key inhibitory relay in the CVLM.
Figures





References
-
- Abercrombie M. Estimation of nuclear population from microtome sections. Anat Rec. 1946;94:239–247. - PubMed
-
- Agarwal SK, Calaresu FR. Monosynaptic connection from caudal to rostral ventrolateral medulla in the baroreceptor reflex pathway. Brain Res. 1991;555:70–74. - PubMed
-
- Aicher SA, Kurucz OS, Reis DJ, Milner TA. Nucleus tractus solitarius efferent terminals synapse on neurons in the caudal ventrolateral medulla that project to the rostral ventrolateral medulla. Brain Res. 1995;693:51–63. - PubMed
-
- Altschuler SM, Bao X, Bieger D, Hopkins DA, Miselis RR. Viscerotopic representation of the upper alimentary tract in the rat: sensory ganglia and nuclei of the solitary and spinal trigeminal tracts. J Comp Neurol. 1989;283:248–268. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
