Onset of feedback reactions underlying vertebrate rod photoreceptor light adaptation
- PMID: 9417133
- PMCID: PMC1887766
- DOI: 10.1085/jgp.111.1.39
Onset of feedback reactions underlying vertebrate rod photoreceptor light adaptation
Abstract
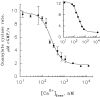
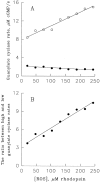
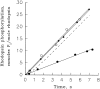
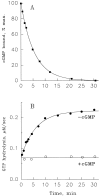
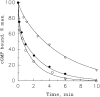
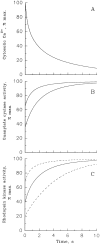
Light adaptation in vertebrate photoreceptors is thought to be mediated through a number of biochemical feedback reactions that reduce the sensitivity of the photoreceptor and accelerate the kinetics of the photoresponse. Ca2+ plays a major role in this process by regulating several components of the phototransduction cascade. Guanylate cyclase and rhodopsin kinase are suggested to be the major sites regulated by Ca2+. Recently, it was proposed that cGMP may be another messenger of light adaptation since it is able to regulate the rate of transducin GTPase and thus the lifetime of activated cGMP phosphodiesterase. Here we report measurements of the rates at which the changes in Ca2+ and cGMP are followed by the changes in the rates of corresponding enzymatic reactions in frog rod outer segments. Our data indicate that there is a temporal hierarchy among reactions that underlie light adaptation. Guanylate cyclase activity and rhodopsin phosphorylation respond to changes in Ca2+ very rapidly, on a subsecond time scale. This enables them to accelerate the falling phase of the flash response and to modulate flash sensitivity during continuous illumination. To the contrary, the acceleration of transducin GTPase, even after significant reduction in cGMP, occurs over several tens of seconds. It is substantially delayed by the slow dissociation of cGMP from the noncatalytic sites for cGMP binding located on cGMP phosphodiesterase. Therefore, cGMP-dependent regulation of transducin GTPase is likely to occur only during prolonged bright illumination.
Figures
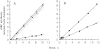

Similar articles
-
cGMP suppresses GTPase activity of a portion of transducin equimolar to phosphodiesterase in frog rod outer segments. Light-induced cGMP decreases as a putative feedback mechanism of the photoresponse.J Biol Chem. 1991 Oct 5;266(28):18530-7. J Biol Chem. 1991. PMID: 1655754
-
Tuning outer segment Ca2+ homeostasis to phototransduction in rods and cones.Adv Exp Med Biol. 2002;514:179-203. doi: 10.1007/978-1-4615-0121-3_11. Adv Exp Med Biol. 2002. PMID: 12596922 Review.
-
Noncatalytic cGMP-binding sites of amphibian rod cGMP phosphodiesterase control interaction with its inhibitory gamma-subunits. A putative regulatory mechanism of the rod photoresponse.J Biol Chem. 1992 Dec 5;267(34):24501-7. J Biol Chem. 1992. PMID: 1332960
-
Mechanism of transducin activation of frog rod photoreceptor phosphodiesterase. Allosteric interactiona between the inhibitory gamma subunit and the noncatalytic cGMP-binding sites.J Biol Chem. 2000 Dec 8;275(49):38611-9. doi: 10.1074/jbc.M004606200. J Biol Chem. 2000. PMID: 10993884
-
Visual excitation and recovery.J Biol Chem. 1991 Jun 15;266(17):10711-4. J Biol Chem. 1991. PMID: 1710212 Review. No abstract available.
Cited by
-
Developmental regulation of calcium-dependent feedback in Xenopus rods.J Gen Physiol. 2004 Nov;124(5):569-85. doi: 10.1085/jgp.200409162. J Gen Physiol. 2004. PMID: 15504902 Free PMC article.
-
Timing is everything: GTPase regulation in phototransduction.Invest Ophthalmol Vis Sci. 2013 Nov 21;54(12):7725-33. doi: 10.1167/iovs.13-13281. Invest Ophthalmol Vis Sci. 2013. PMID: 24265205 Free PMC article. Review.
-
Kinetics of turn-offs of frog rod phototransduction cascade.J Gen Physiol. 2008 Nov;132(5):587-604. doi: 10.1085/jgp.200810034. J Gen Physiol. 2008. PMID: 18955597 Free PMC article.
-
Enzymatic relay mechanism stimulates cyclic GMP synthesis in rod photoresponse: biochemical and physiological study in guanylyl cyclase activating protein 1 knockout mice.PLoS One. 2012;7(10):e47637. doi: 10.1371/journal.pone.0047637. Epub 2012 Oct 17. PLoS One. 2012. PMID: 23082185 Free PMC article.
-
Direct allosteric regulation between the GAF domain and catalytic domain of photoreceptor phosphodiesterase PDE6.J Biol Chem. 2008 Oct 31;283(44):29699-705. doi: 10.1074/jbc.M803948200. Epub 2008 Sep 8. J Biol Chem. 2008. PMID: 18779324 Free PMC article.
References
-
- Ames JB, Porumb T, Tanaka T, Ikura M, Stryer L. Amino-terminal myristoylation induces cooperative calcium binding to recoverin. J Biol Chem. 1995;270:4526–4533. - PubMed
-
- Aparicio JG, Applebury ML. The photoreceptor guanylate cyclase is an autophosphorylating protein kinase. J Biol Chem. 1996;271:27083–27089. - PubMed
-
- Arshavsky VY, Dumke CL, Bownds MD. Non-catalytic cGMP binding sites of amphibian rod cGMP phosphodiesterase control interaction with its inhibitory gamma-subunits—a putative regulatory mechanism of the rod photoresponse. J Biol Chem. 1992;267:24501–24507. - PubMed
-
- Arshavsky VY, Gray-Keller MP, Bownds MD. cGMP suppresses GTPase activity of a portion of transducin equimolar to phosphodiesterase in frog rod outer segments. Light-induced cGMP decreases as a putative feedback mechanism of the photoresponse. J Biol Chem. 1991;266:18530–18537. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

