Properties of an inwardly rectifying ATP-sensitive K+ channel in the basolateral membrane of renal proximal tubule
- PMID: 9417141
- PMCID: PMC1887768
- DOI: 10.1085/jgp.111.1.139
Properties of an inwardly rectifying ATP-sensitive K+ channel in the basolateral membrane of renal proximal tubule
Abstract
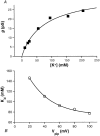
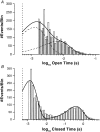
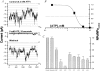
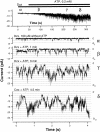
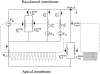
The potassium conductance of the basolateral membrane (BLM) of proximal tubule cells is a critical regulator of transport since it is the major determinant of the negative cell membrane potential and is necessary for pump-leak coupling to the Na+,K+-ATPase pump. Despite this pivotal physiological role, the properties of this conductance have been incompletely characterized, in part due to difficulty gaining access to the BLM. We have investigated the properties of this BLM K+ conductance in dissociated, polarized Ambystoma proximal tubule cells. Nearly all seals made on Ambystoma cells contained inward rectifier K+ channels (gammaslope, in = 24.5 +/- 0.6 pS, gammachord, out = 3.7 +/- 0.4 pS). The rectification is mediated in part by internal Mg2+. The open probability of the channel increases modestly with hyperpolarization. The inward conducting properties are described by a saturating binding-unbinding model. The channel conducts Tl+ and K+, but there is no significant conductance for Na+, Rb+, Cs+, Li+, NH4+, or Cl-. The channel is inhibited by barium and the sulfonylurea agent glibenclamide, but not by tetraethylammonium. Channel rundown typically occurs in the absence of ATP, but cytosolic addition of 0. 2 mM ATP (or any hydrolyzable nucleoside triphosphate) sustains channel activity indefinitely. Phosphorylation processes alone fail to sustain channel activity. Higher doses of ATP (or other nucleoside triphosphates) reversibly inhibit the channel. The K+ channel opener diazoxide opens the channel in the presence of 0.2 mM ATP, but does not alleviate the inhibition of millimolar doses of ATP. We conclude that this K+ channel is the major ATP-sensitive basolateral K+ conductance in the proximal tubule.
Figures




Similar articles
-
Regulation of an inwardly rectifying ATP-sensitive K+ channel in the basolateral membrane of renal proximal tubule.J Gen Physiol. 1998 Jan;111(1):161-80. doi: 10.1085/jgp.111.1.161. J Gen Physiol. 1998. PMID: 9417142 Free PMC article.
-
A mechanogated nonselective cation channel in proximal tubule that is ATP sensitive.Am J Physiol Renal Physiol. 2002 Jul;283(1):F93-F104. doi: 10.1152/ajprenal.00239.2001. Am J Physiol Renal Physiol. 2002. PMID: 12060591
-
An intracellular ATP-activated, calcium-permeable conductance on the basolateral membrane of single renal proximal tubule cells isolated from Rana temporaria.J Physiol. 2000 Mar 1;523 Pt 2(Pt 2):301-11. doi: 10.1111/j.1469-7793.2000.00301.x. J Physiol. 2000. PMID: 10699076 Free PMC article.
-
ATP-sensitive K+ channels in the kidney.Naunyn Schmiedebergs Arch Pharmacol. 1996 Aug-Sep;354(3):213-25. doi: 10.1007/BF00171051. Naunyn Schmiedebergs Arch Pharmacol. 1996. PMID: 8878050 Review.
-
Regulation of renal proximal tubule basolateral potassium channels.Prog Clin Biol Res. 1990;334:231-49. Prog Clin Biol Res. 1990. PMID: 2408069 Review.
Cited by
-
Phosphorylation regulates an inwardly rectifying ATP-sensitive K(+)- conductance in proximal tubule cells of frog kidney.J Membr Biol. 2005 Oct;207(3):161-7. doi: 10.1007/s00232-005-0811-2. J Membr Biol. 2005. PMID: 16550487
-
Interaction between Calcineurin and Ca/Calmodulin Kinase-II in Modulating Cellular Functions.Enzyme Res. 2011;2011:587359. doi: 10.4061/2011/587359. Epub 2011 May 30. Enzyme Res. 2011. PMID: 21687603 Free PMC article.
-
Glibenclamide depletes ATP in renal proximal tubular cells by interfering with mitochondrial metabolism.Br J Pharmacol. 2005 Aug;145(8):1069-75. doi: 10.1038/sj.bjp.0706275. Br J Pharmacol. 2005. PMID: 15912128 Free PMC article.
-
Epithelial transport in The Journal of General Physiology.J Gen Physiol. 2017 Oct 2;149(10):897-909. doi: 10.1085/jgp.201711828. Epub 2017 Sep 20. J Gen Physiol. 2017. PMID: 28931633 Free PMC article. Review.
-
Sulphonylurea drugs reduce hypoxic damage in the isolated perfused rat kidney.Br J Pharmacol. 2000 Aug;130(7):1678-84. doi: 10.1038/sj.bjp.0703469. Br J Pharmacol. 2000. PMID: 10928974 Free PMC article.
References
-
- Aguilar-Bryan L, Nichols CG, Wechsler SW, Clement JP, IV, Boyd AE, III, Gonzalez G, Herrera-Sosa H, Nguy K, Bryan J, Nelson DA. Cloning of the beta cell high-affinity sulfonylurea receptor: a regulator of insulin secretion. Science. 1995;268:423–426. - PubMed
-
- Ashcroft SJ, Ashcroft FM. Properties and functions of ATP-sensitive K-channels. Cell Signal. 1990;2:197–214. - PubMed
-
- Beck JS, Hurst AM, Lapointe JY, Laprade R. Regulation of basolateral K channels in proximal tubule studied during continuous microperfusion. Am J Physiol. 1993;264:F496–F501. - PubMed
-
- Beck JS, Breton S, Mairbäurl H, Laprade R, Giebisch G. Relationship between sodium transport and intracellular ATP in isolated perfused rabbit proximal convoluted tubule. Am J Physiol. 1991;261:F634–F639. - PubMed

