Epstein-Barr virus lacking glycoprotein gp42 can bind to B cells but is not able to infect
- PMID: 9420211
- PMCID: PMC109360
- DOI: 10.1128/JVI.72.1.158-163.1998
Epstein-Barr virus lacking glycoprotein gp42 can bind to B cells but is not able to infect
Abstract
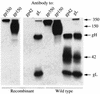
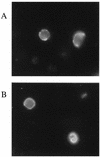
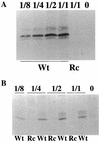
The Epstein-Barr virus gH-gL complex includes a third glycoprotein, gp42, which is the product of the BZLF2 open reading frame (ORF). gp42 has been implicated as critical to infection of the B lymphocyte by virtue of its interaction with HLA class II on the B-cell surface. A neutralizing antibody that reacts with gp42 inhibits virus-cell fusion and blocks binding of gp42 to HLA class II; antibody to HLA class II can inhibit infection, and B cells that lack HLA class II can only be infected if HLA class II expression is restored. To confirm whether gp42 is an essential component of the virion, we derived a recombinant virus with a selectable marker inserted into the BZLF2 ORF to interrupt expression of the protein. A complex of gH and gL was expressed by the recombinant virus in the absence of gp42. Recombinant virus egressed from the cell normally and could bind to receptor-positive cells. It had, however, lost the ability to infect or transform B lymphocytes. Treatment with polyethylene glycol restored the infectivity of recombinant virus, confirming that gp42 is essential for penetration of the B-cell membrane.
Figures

Similar articles
-
Epstein-Barr virus gH is essential for penetration of B cells but also plays a role in attachment of virus to epithelial cells.J Virol. 2000 Jul;74(14):6324-32. doi: 10.1128/jvi.74.14.6324-6332.2000. J Virol. 2000. PMID: 10864642 Free PMC article.
-
Epstein-Barr virus uses different complexes of glycoproteins gH and gL to infect B lymphocytes and epithelial cells.J Virol. 1998 Jul;72(7):5552-8. doi: 10.1128/JVI.72.7.5552-5558.1998. J Virol. 1998. PMID: 9621012 Free PMC article.
-
The Epstein-Barr virus (EBV) BZLF2 gene product associates with the gH and gL homologs of EBV and carries an epitope critical to infection of B cells but not of epithelial cells.J Virol. 1995 Jul;69(7):3987-94. doi: 10.1128/JVI.69.7.3987-3994.1995. J Virol. 1995. PMID: 7539502 Free PMC article.
-
Structural and Mechanistic Insights into the Tropism of Epstein-Barr Virus.Mol Cells. 2016 Apr 30;39(4):286-91. doi: 10.14348/molcells.2016.0066. Epub 2016 Apr 6. Mol Cells. 2016. PMID: 27094060 Free PMC article. Review.
-
[The entry of Epstein-Barr virus into B lymphocytes and epithelial cells during infection].Bing Du Xue Bao. 2014 Jul;30(4):476-82. Bing Du Xue Bao. 2014. PMID: 25272606 Review. Chinese.
Cited by
-
A gH/gL-encoding replicon vaccine elicits neutralizing antibodies that protect humanized mice against EBV challenge.NPJ Vaccines. 2024 Jun 26;9(1):120. doi: 10.1038/s41541-024-00907-y. NPJ Vaccines. 2024. PMID: 38926438 Free PMC article.
-
The amino terminus of Epstein-Barr virus glycoprotein gH is important for fusion with epithelial and B cells.J Virol. 2005 Oct;79(19):12408-15. doi: 10.1128/JVI.79.19.12408-12415.2005. J Virol. 2005. PMID: 16160168 Free PMC article.
-
Epstein-Barr viruses that express a CD21 antibody provide evidence that gp350's functions extend beyond B-cell surface binding.J Virol. 2010 Jan;84(2):1139-47. doi: 10.1128/JVI.01953-09. Epub 2009 Nov 4. J Virol. 2010. PMID: 19889766 Free PMC article.
-
Epithelial cell infection by Epstein-Barr virus.FEMS Microbiol Rev. 2019 Nov 1;43(6):674-683. doi: 10.1093/femsre/fuz023. FEMS Microbiol Rev. 2019. PMID: 31584659 Free PMC article.
-
The genes encoding the gCIII complex of human cytomegalovirus exist in highly diverse combinations in clinical isolates.J Virol. 2002 Nov;76(21):10841-8. doi: 10.1128/jvi.76.21.10841-10848.2002. J Virol. 2002. PMID: 12368327 Free PMC article.
References
-
- Agius C T, Studdert M J. Equine herpesvirus 2 and 5: comparisons with other members of the subfamily gammaherpesvirinae. Adv Virus Res. 1994;44:357–379. - PubMed
-
- Ausubel F M, Brent R, Kingston R E, Moore D D, Seidman J G, Smith J A, Struhl K, editors. Current protocols in molecular biology. New York, N.Y: John Wiley and Sons, Inc.; 1997.
-
- Baer R, Bankier A T, Biggin M D, Deininger P L, Farrell P J, Gibson T J, Hatfull G, Satchwell S C, Seguin C, Tuffnell P S, Barrell B G. DNA sequence and expression of the B95-8 Epstein-Barr virus genome. Nature (London) 1984;310:207–211. - PubMed
-
- Desai P J, Schaffer P A, Minson A C. Excretion of non-infectious virus particles lacking glycoprotein H by a temperature-sensitive mutant of herpes simplex virus type 1: evidence that gH is essential for virion infectivity. J Gen Virol. 1988;69:1147–1156. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

