The glutamate-rich protein (GLURP) of Plasmodium falciparum is a target for antibody-dependent monocyte-mediated inhibition of parasite growth in vitro
- PMID: 9423833
- PMCID: PMC107852
- DOI: 10.1128/IAI.66.1.11-17.1998
The glutamate-rich protein (GLURP) of Plasmodium falciparum is a target for antibody-dependent monocyte-mediated inhibition of parasite growth in vitro
Abstract
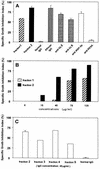
Monocyte-dependent as well as direct inhibitory effects of antimalarial antibodies point toward antigens accessible at the time of merozoite release as targets for biologically active antibodies capable of mediating protection against Plasmodium falciparum. The glutamate-rich protein (GLURP), being an antigen associated with mature schizont-infected erythrocytes, was therefore the object of the present investigation, in which we analyzed whether anti-GLURP antibodies can either interfere directly with merozoite invasion or act indirectly by promoting a monocyte-dependent growth inhibition, antibody-dependent cellular inhibition. GLURP-specific human immunoglobulin G (IgG) antibodies, from pooled IgG of healthy Liberian adults who were clinically immune to malaria, were purified by affinity chromatography on columns containing R0 (N-terminal nonrepetitive region of GLURP) or R2 (C-terminal repetitive region of GLURP) recombinant protein or synthetic peptides as ligands. Analysis of the pattern of reactivity of highly purified anti-GLURP antibodies led to the definition of at least four B-cell epitopes. One epitope was specific for R0, two were specific for R2, and the fourth displayed cross-reactivity between R0 and R2. None of the purified IgG antibodies had direct invasion-inhibitory effects, even at high concentrations. In contrast, when allowed to cooperate with monocytes, all anti-GLURP IgG preparations mediated a strong monocyte-dependent parasite growth inhibition in a dose-dependent manner.
Figures

Similar articles
-
Identification of a major B-cell epitope of the Plasmodium falciparum glutamate-rich protein (GLURP), targeted by human antibodies mediating parasite killing.Vaccine. 2000 Sep 15;19(2-3):204-12. doi: 10.1016/s0264-410x(00)00181-x. Vaccine. 2000. PMID: 10930674
-
Antibodies against the Plasmodium falciparum glutamate-rich protein from naturally exposed individuals living in a Brazilian malaria-endemic area can inhibit in vitro parasite growth.Mem Inst Oswaldo Cruz. 2011 Aug;106 Suppl 1:34-43. doi: 10.1590/s0074-02762011000900005. Mem Inst Oswaldo Cruz. 2011. PMID: 21881755
-
Antibody Correlates of Protection from Clinical Plasmodium falciparum Malaria in an Area of Low and Unstable Malaria Transmission.Am J Trop Med Hyg. 2020 Dec;103(6):2174-2182. doi: 10.4269/ajtmh.18-0805. Epub 2020 Oct 27. Am J Trop Med Hyg. 2020. PMID: 33124533 Free PMC article. Review.
-
Antigenicity and immunogenicity of recombinant glutamate-rich protein of Plasmodium falciparum expressed in Escherichia coli.Clin Diagn Lab Immunol. 1995 Jan;2(1):30-4. doi: 10.1128/cdli.2.1.30-34.1995. Clin Diagn Lab Immunol. 1995. PMID: 7719909 Free PMC article.
-
Glutamate rich Plasmodium falciparum antigen (GLURP).Parassitologia. 1993 Jul;35 Suppl:47-50. Parassitologia. 1993. PMID: 8233611 Review.
Cited by
-
Association between protection against clinical malaria and antibodies to merozoite surface antigens in an area of hyperendemicity in Myanmar: complementarity between responses to merozoite surface protein 3 and the 220-kilodalton glutamate-rich protein.Infect Immun. 2004 Jan;72(1):247-52. doi: 10.1128/IAI.72.1.247-252.2004. Infect Immun. 2004. PMID: 14688102 Free PMC article.
-
Variation in the immune responses against Plasmodium falciparum merozoite surface protein-1 and apical membrane antigen-1 in children residing in the different epidemiological strata of malaria in Cameroon.Malar J. 2017 Nov 9;16(1):453. doi: 10.1186/s12936-017-2105-4. Malar J. 2017. PMID: 29121929 Free PMC article.
-
Expression, Purification and Characterization of GMZ2'.10C, a Complex Disulphide-Bonded Fusion Protein Vaccine Candidate against the Asexual and Sexual Life-Stages of the Malaria-Causing Plasmodium falciparum Parasite.Pharm Res. 2017 Sep;34(9):1970-1983. doi: 10.1007/s11095-017-2208-1. Epub 2017 Jun 23. Pharm Res. 2017. PMID: 28646324
-
Antigenic characterization of an intrinsically unstructured protein, Plasmodium falciparum merozoite surface protein 2.Infect Immun. 2012 Dec;80(12):4177-85. doi: 10.1128/IAI.00665-12. Epub 2012 Sep 10. Infect Immun. 2012. PMID: 22966050 Free PMC article.
-
Specificity of the protective antibody response to apical membrane antigen 1.Infect Immun. 2001 May;69(5):3286-94. doi: 10.1128/IAI.69.5.3286-3294.2001. Infect Immun. 2001. PMID: 11292751 Free PMC article.
References
-
- Anders R F. Multiple cross-reactivities amongst antigens of Plasmodium falciparum impair the development of protective immunity against malaria. Parasite Immunol. 1986;8:529–539. - PubMed
-
- Borre M B, Dziegiel M, Hogh B, Petersen E, Rieneck K, Riley E, Meis J F, Aikawa M, Nakamura K, Harada M, Wind A, Jakobsen P H, Cowland J, Jepsen S, Axelsen N H, Vuust J. Primary structure and localization of a conserved immunogenic Plasmodium falciparum glutamate rich protein (GLURP) expressed in both the preerythrocytic and erythrocytic stages of the vertebrate life cycle. Mol Biochem Parasitol. 1991;49:119–132. - PubMed
-
- Bottius E, Guanzirolli A, Trape J-F, Rogier C, Konate L, Druilhe P. Malaria: even more chronic in nature than previously thought; evidence for submicroscopic parasitaemia detectable by polymerase chain reaction. Trans R Soc Trop Med Hyg. 1996;90:15–19. - PubMed
-
- Bouharoun-Tayoun H, Attanah P, Sabchareon A, Chongsuphajaisiddhi T, Druilhe P. Antibodies that protect humans against Plasmodium falciparum blood stages do not on their own inhibit parasite growth and invasion in vitro, but act in cooperation with monocytes. J Exp Med. 1990;172:1633–1641. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

