Molecular mechanisms of lymphocyte homing to peripheral lymph nodes
- PMID: 9432978
- PMCID: PMC2212097
- DOI: 10.1084/jem.187.2.205
Molecular mechanisms of lymphocyte homing to peripheral lymph nodes
Abstract
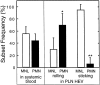
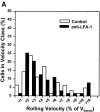
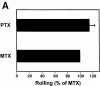
To characterize the adhesion cascade that directs lymphocyte homing to peripheral lymph nodes (PLNs), we investigated the molecular mechanisms of lymphocyte interactions with the microvasculature of subiliac lymph nodes. We found that endogenous white blood cells and adoptively transferred lymph node lymphocytes (LNCs) tethered and rolled in postcapillary high endothelial venules (HEVs) and to a lesser extent in collecting venules. Similarly, firm arrest occurred nearly exclusively in the paracortical HEVs. Endogenous polymorphonuclear (PMNs) and mononuclear leukocytes (MNLs) attached and rolled in HEVs at similar frequencies, but only MNLs arrested suggesting that the events downstream of primary rolling interactions critically determine the specificity of lymphocyte recruitment. Antibody inhibition studies revealed that L-selectin was responsible for attachment and rolling of LNCs, and that LFA-1 was essential for sticking. LFA-1-dependent arrest was also abolished by pertussis toxin, implicating a requirement for G alpha i--protein-linked signaling. alpha 4 integrins, which play a critical role in lymphocyte homing to Peyer's Patches, made no significant contribution to attachment, rolling, or sticking in resting PLNs. Velocity analysis of interacting LNCs revealed no detectable contribution by LFA-1 to rolling. Taken together, our results suggest that lymphocyte- HEV interactions within PLNs are almost exclusively initiated by L-selectin followed by a G protein-coupled lymphocyte-specific activation event and activation-induced engagement of LFA-1. These events constitute a unique adhesion cascade that dictates the specificity of lymphocyte homing to PLNs.
Figures
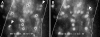









References
-
- Gowans JL, Knight EJ. The route of re-circulation of lymphocytes in the rat. Proc R Soc Lond B Biol. 1964;159:257–282. - PubMed
-
- Butcher EC, Scollay RG, Weissman IL. Organ specificity of lymphocyte migration: mediation by highly selective lymphocyte interaction with organ-specific determinants on high endothelial venules. Eur J Immunol. 1980;10:556–561. - PubMed
-
- Butcher EC, Picker LJ. Lymphocyte homing and homeostasis. Science. 1996;272:60–66. - PubMed
-
- Butcher EC. Leukocyte-endothelial cell recognition: three (or more) steps to specificity and diversity. Cell. 1991;67:1033–1036. - PubMed
-
- Shimizu Y, Newman W, Tanaka Y, Shaw S. Lymphocyte interactions with endothelial cells. Immunol Today. 1992;13:106–112. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

