Der3p/Hrd1p is required for endoplasmic reticulum-associated degradation of misfolded lumenal and integral membrane proteins
- PMID: 9437001
- PMCID: PMC25244
- DOI: 10.1091/mbc.9.1.209
Der3p/Hrd1p is required for endoplasmic reticulum-associated degradation of misfolded lumenal and integral membrane proteins
Abstract
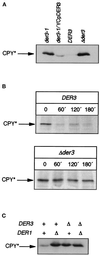
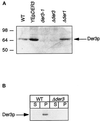
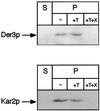
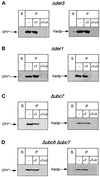
We have studied components of the endoplasmic reticulum (ER) proofreading and degradation system in the yeast Saccharomyces cerevisiae. Using a der3-1 mutant defective in the degradation of a mutated lumenal protein, carboxypeptidase yscY (CPY*), a gene was cloned which encodes a 64-kDa protein of the ER membrane. Der3p was found to be identical with Hrd1p, a protein identified to be necessary for degradation of HMG-CoA reductase. Der3p contains five putative transmembrane domains and a long hydrophilic C-terminal tail containing a RING-H2 finger domain which is oriented to the ER lumen. Deletion of DER3 leads to an accumulation of CPY* inside the ER due to a complete block of its degradation. In addition, a DER3 null mutant allele suppresses the temperature-dependent growth phenotype of a mutant carrying the sec61-2 allele. This is accompanied by the stabilization of the Sec61-2 mutant protein. In contrast, overproduction of Der3p is lethal in a sec61-2 strain at the permissive temperature of 25 degrees C. A mutant Der3p lacking 114 amino acids of the lumenal tail including the RING-H2 finger domain is unable to mediate degradation of CPY* and Sec61-2p. We propose that Der3p acts prior to retrograde transport of ER membrane and lumenal proteins to the cytoplasm where they are subject to degradation via the ubiquitin-proteasome system. Interestingly, in ubc6-ubc7 double mutants, CPY* accumulates in the ER, indicating the necessity of an intact cytoplasmic proteolysis machinery for retrograde transport of CPY*. Der3p might serve as a component programming the translocon for retrograde transport of ER proteins, or it might be involved in recognition through its lumenal RING-H2 motif of proteins of the ER that are destined for degradation.
Figures





References
-
- Amitay R, Shachar I, Rabinovich, Haimovich E, J, Bar-Nun S. Degradation of secretory immunoglobulin M in B lymphocytes occurs in a post endoplasmic compartment and is mediated by a cysteine protease. J Biol Chem. 1992;267:20964–20700. - PubMed
-
- Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K. Current Protocols in Molecular Biology. New York: Greene Publishing Associates and Wiley Interscience; 1992.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

