Differential tropism and chemokine receptor expression of human immunodeficiency virus type 1 in neonatal monocytes, monocyte-derived macrophages, and placental macrophages
- PMID: 9445034
- PMCID: PMC124612
- DOI: 10.1128/JVI.72.2.1334-1344.1998
Differential tropism and chemokine receptor expression of human immunodeficiency virus type 1 in neonatal monocytes, monocyte-derived macrophages, and placental macrophages
Abstract
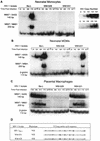
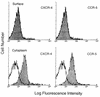
Laboratory-adapted (LA) macrophage-tropic (M-tropic) human immunodeficiency virus type 1 (HIV-1) isolates (e.g., HIV-1(Ba-L)) and low-passage primary (PR) isolates differed markedly in tropism for syngeneic neonatal monocytes, monocyte-derived macrophages (MDMs), and placental macrophages (PMs). Newly adherent neonatal monocytes and cultured PMs were highly refractory to infection with PR HIV-1 isolates yet were permissive for LA M-tropic isolates. Day 4 MDMs were also permissive for LA M-tropic isolates and additionally, were permissive for over half the PR isolates tested. Qualitative differences in PR HIV-1 infection of monocytes/MDMs could not be correlated with CD4 levels alone, and in all three cell types the block to PR HIV-1 strain replication preceded reverse transcription. Neonatal monocyte susceptibility to PR HIV-1 strains correlated with increasing CCR-5 expression during maturation. CCR-5 could not be detected on newly adherent (day 1) neonatal monocytes, in contrast to adult monocytes (H. Naif et al., J. Virol. 72:830-836, 1998), but was readily detectable after 4 to 7 days of culture. However, moderate CCR-5 mRNA levels were present in day 1 neonatal monocytes and remained constant during monocyte maturation. CCR-5 was not detectable on the surface of PMs, yet the receptor was present within permeabilized cells. Notably, two brain-derived PR HIV-1 isolates from a single patient, differing in their V3 loops, were discordant in their abilities to infect neonatal monocytes/MDMs and PMs, yet both isolates could infect newly adherent adult monocytes. Together these data strongly suggest that LA HIV-1 isolates are able to infect neonatal monocytes at earlier stages of maturation and lower-level expression of CCR-5 than PR isolates. The differences between neonatal and adult monocytes in susceptibility to PR isolates may also be related to the level of CCR-5 expression.
Figures







References
-
- Alkhatib G, Combadiere C, Broder C C, Feng Y, Kennedy P E, Murphy P M, Berger E A. CC CKR5: a RANTES, MIP-1α, MIP-1β receptor as a fusion cofactor for macrophage-tropic HIV-1. Science. 1996;272:1955–1958. - PubMed
-
- Chandwani S, Greco M, Mittal K, Antoine C, Kraninski K, Borkowsky W. Pathology and human immunodeficiency virus expression in placentas of seropositive women. J Infect Dis. 1991;163:1134–1138. - PubMed
-
- Chang J, Li S, Naif H, Cunningham A L. The magnitude of HIV replication in monocytes and macrophages is influenced by environmental conditions, viral strain, and host cells. J Leukocyte Biol. 1994;56:230–235. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

