Activation of Shaker potassium channels. III. An activation gating model for wild-type and V2 mutant channels
- PMID: 9450946
- PMCID: PMC2222769
- DOI: 10.1085/jgp.111.2.313
Activation of Shaker potassium channels. III. An activation gating model for wild-type and V2 mutant channels
Abstract
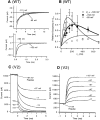
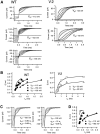
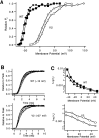
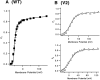
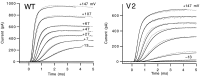
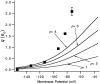
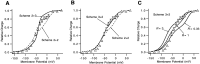
A functional kinetic model is developed to describe the activation gating process of the Shaker potassium channel. The modeling in this paper is constrained by measurements described in the preceding two papers, including macroscopic ionic and gating currents and single channel ionic currents. These data were obtained from the normally activating wild-type channel as well as a mutant channel V2, in which the leucine at position 382 has been mutated to a valine. Different classes of models that incorporate Shaker's symmetrical tetrameric structure are systematically examined. Many simple gating models are clearly inadequate, but a model that can account for all of the qualitative features of the data has the channel open after its four subunits undergo three transitions in sequence, and two final transitions that reflect the concerted action of the four subunits. In this model, which we call Scheme 3+2', the channel can also close to several states that are not part of the activation path. Channel opening involves a large total charge movement (10.8 e0), which is distributed among a large number of small steps each with rather small charge movements (between 0.6 and 1.05 e0). The final two transitions are different from earlier steps by having slow backward rates. These steps confer a cooperative mechanism of channel opening at Shaker's activation voltages. In the context of Scheme 3+2', significant effects of the V2 mutation are limited to the backward rates of the final two transitions, implying that L382 plays an important role in the conformational stability of the final two states.
Figures











Similar articles
-
Activation of Shaker potassium channels. II. Kinetics of the V2 mutant channel.J Gen Physiol. 1998 Feb;111(2):295-311. doi: 10.1085/jgp.111.2.295. J Gen Physiol. 1998. PMID: 9450945 Free PMC article.
-
Activation of shaker potassium channels. I. Characterization of voltage-dependent transitions.J Gen Physiol. 1998 Feb;111(2):271-94. doi: 10.1085/jgp.111.2.271. J Gen Physiol. 1998. PMID: 9450944 Free PMC article.
-
Determinants of voltage-dependent gating and open-state stability in the S5 segment of Shaker potassium channels.J Gen Physiol. 1999 Aug;114(2):215-42. doi: 10.1085/jgp.114.2.215. J Gen Physiol. 1999. PMID: 10435999 Free PMC article.
-
Gating of voltage-dependent potassium channels.Prog Biophys Mol Biol. 2001;75(3):165-99. doi: 10.1016/s0079-6107(01)00006-2. Prog Biophys Mol Biol. 2001. PMID: 11376798 Review.
-
A historical biophysical dogma vs. an understanding of the structure and function of voltage-gated tetrameric ion channels. A review.Biochim Biophys Acta Biomembr. 2022 Dec 1;1864(12):184046. doi: 10.1016/j.bbamem.2022.184046. Epub 2022 Sep 10. Biochim Biophys Acta Biomembr. 2022. PMID: 36096197 Review.
Cited by
-
Molecular mechanism of TRP channels.Compr Physiol. 2013 Jan;3(1):221-42. doi: 10.1002/cphy.c120001. Compr Physiol. 2013. PMID: 23720286 Free PMC article. Review.
-
Basis for allosteric open-state stabilization of voltage-gated potassium channels by intracellular cations.J Gen Physiol. 2012 Nov;140(5):495-511. doi: 10.1085/jgp.201210823. Epub 2012 Oct 15. J Gen Physiol. 2012. PMID: 23071269 Free PMC article.
-
Molecular dynamics simulations of voltage-gated cation channels: insights on voltage-sensor domain function and modulation.Front Pharmacol. 2012 May 25;3:97. doi: 10.3389/fphar.2012.00097. eCollection 2012. Front Pharmacol. 2012. PMID: 22654756 Free PMC article.
-
Voltage Sensing in Membranes: From Macroscopic Currents to Molecular Motions.J Membr Biol. 2015 Jun;248(3):419-30. doi: 10.1007/s00232-015-9805-x. Epub 2015 May 14. J Membr Biol. 2015. PMID: 25972106 Free PMC article. Review.
-
Activation properties of Kv4.3 channels: time, voltage and [K+]o dependence.J Physiol. 2004 Jun 15;557(Pt 3):705-17. doi: 10.1113/jphysiol.2003.058578. Epub 2004 Mar 5. J Physiol. 2004. PMID: 15004209 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

