Comparison of intranasal and intramuscular immunization against human immunodeficiency virus type 1 with a DNA-monophosphoryl lipid A adjuvant vaccine
- PMID: 9453648
- PMCID: PMC107976
- DOI: 10.1128/IAI.66.2.823-826.1998
Comparison of intranasal and intramuscular immunization against human immunodeficiency virus type 1 with a DNA-monophosphoryl lipid A adjuvant vaccine
Abstract
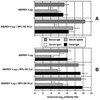
We compared immune responses to intranasal and intramuscular DNA vaccinations against human immunodeficiency virus type 1 with monophosphoryl lipid A (MPL) used as an adjuvant. Both routes of vaccination resulted in similar levels of cell-mediated immunity, but the intestinal secretory immunoglobulin A response was higher following intranasal immunization than after intramuscular immunization. MPL demonstrated its adjuvanticity in vaccination by both routes.
Figures

References
-
- Ada G L, McElrath M J. HIV type 1 vaccine-induced cytotoxic T cell responses: potential role in vaccine efficacy. AIDS Res Hum Retroviruses. 1997;13:205–210. - PubMed
-
- Bukawa H, Sekigawa K, Hamajima K, Fukushima J, Yamada Y, Kiyono H, Okuda K. Neutralization of HIV-1 by secretory IgA induced by oral immunization with a new macromolecular multicomponent peptide vaccine candidate. Nat Med. 1995;1:681–685. - PubMed
-
- Donnelly J J, Ulmer J B, Shiver J W, Liu M A. DNA vaccines. Annu Rev Immunol. 1997;15:617–648. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

