Titin extensibility in situ: entropic elasticity of permanently folded and permanently unfolded molecular segments
- PMID: 9472037
- PMCID: PMC2141751
- DOI: 10.1083/jcb.140.4.853
Titin extensibility in situ: entropic elasticity of permanently folded and permanently unfolded molecular segments
Abstract
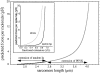
Titin (also known as connectin) is a giant protein that spans half of the striated muscle sarcomere. In the I-band titin extends as the sarcomere is stretched, developing what is known as passive force. The I-band region of titin contains tandem Ig segments (consisting of serially linked immunoglobulin-like domains) with the unique PEVK segment in between (Labeit, S., and B. Kolmerer. 1995. Science. 270:293-296). Although the tandem Ig and PEVK segments have been proposed to behave as stiff and compliant springs, respectively, precise experimental testing of the hypothesis is still needed. Here, sequence-specific antibodies were used to mark the ends of the tandem Ig and PEVK segments. By following the extension of the segments as a function of sarcomere length (SL), their respective contributions to titin's elastic behavior were established. In slack sarcomeres (approximately 2.0 micron) the tandem Ig and PEVK segments were contracted. Upon stretching sarcomeres from approximately 2.0 to 2.7 micron, the "contracted" tandem Ig segments straightened while their individual Ig domains remained folded. When sarcomeres were stretched beyond approximately 2.7 micron, the tandem Ig segments did not further extend, instead PEVK extension was now dominant. Modeling tandem Ig and PEVK segments as entropic springs with different bending rigidities (Kellermayer, M., S. Smith, H. Granzier, and C. Bustamante. 1997. Science. 276:1112-1116) indicated that in the physiological SL range (a) the Ig-like domains of the tandem Ig segments remain folded and (b) the PEVK segment behaves as a permanently unfolded polypeptide. Our model provides a molecular basis for the sequential extension of titin's different segments. Initially, the tandem Ig segments extend at low forces due to their high bending rigidity. Subsequently, extension of the PEVK segment occurs only upon reaching sufficiently high external forces due to its low bending rigidity. The serial linking of tandem Ig and PEVK segments with different bending rigidities provides a unique passive force-SL relation that is not achievable with a single elastic segment.
Figures


References
-
- Bustamante C, Marko J, Siggia E, Smith S. Entropic elasticity of λ-phage DNA. Science. 1994;265:1599–1600. - PubMed
-
- Cantor, C., and P. Schimmel. 1980. The Behavior of Biological Macromolecules. Freeman and Company, San Francisco. 849–1371.
-
- Daughdrill G, Chadsey M, Karlinsey J, Hughes K, Dahlquist F. The C-terminal half of anti-sigma factor, FlgM, becomes structured when bound to its target, σ. Nat Struct Biol. 1997;4:285–291. - PubMed
-
- Fürst D, Gautel M. The anatomy of a molecular giant: how the sarcomeric cytoskeleton is assembled from molecules of the immunoglobulin superfamily. J Mol Cell Cardiol. 1995;27:951–959. - PubMed
-
- Gautel M, Goulding D. A molecular map of titin/connectin elasticity reveals two different mechanisms acting in series. FEBS Lett. 1996;385:11–14. - PubMed

