B lymphocyte chemotaxis regulated in association with microanatomic localization, differentiation state, and B cell receptor engagement
- PMID: 9480985
- PMCID: PMC2212170
- DOI: 10.1084/jem.187.5.753
B lymphocyte chemotaxis regulated in association with microanatomic localization, differentiation state, and B cell receptor engagement
Abstract
Migration of mature B lymphocytes within secondary lymphoid organs and recirculation between these sites are thought to allow B cells to obtain T cell help, to undergo somatic hypermutation, to differentiate into effector cells, and to home to sites of antibody production. The mechanisms that direct migration of B lymphocytes are unknown, but there is evidence that G protein-coupled receptors, and possibly chemokine receptors, may be involved. Stromal cell- derived factor (SDF)-1alpha is a CXC chemokine previously characterized as an efficacious chemoattractant for T lymphocytes and monocytes in peripheral blood. Here we show with purified tonsillar B cells that SDF-1alpha also attracts naive and memory, but not germinal center (GC) B lymphocytes. Furthermore, GC B cells could be converted to respond to SDF-1alpha by in vitro differentiation into memory B lymphocytes. Conversely, the migratory response in naive and memory B cells was significantly reduced after B cell receptor engagement and CD40 signaling. The receptor for SDF-1, CXC chemokine receptor 4 (CXCR4), was found to be expressed on responsive as well as unresponsive B cell subsets, but was more rapidly downregulated on responsive cells by ligand. Finally, messenger RNA for SDF-1 was detected by in situ hybridization in a layer of cells surrounding the GC. These findings show that responsiveness to the chemoattractant SDF-1alpha is regulated during B lymphocyte activation, and correlates with positioning of B lymphocytes within a secondary lymphoid organ.
Figures
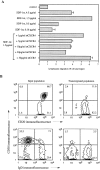


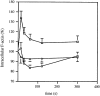
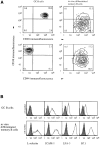





References
-
- Nieuwenhuis P, Ford WL. Comparative migration of B- and T-lymphocytes in the rat spleen and lymph-nodes. Cell Immunol. 1976;23:254–267. - PubMed
-
- MacLennan ICM. Germinal centers. Annu Rev Immunol. 1994;12:117–139. - PubMed
-
- Rajewsky K. Clonal selection and learning in the antibody system. Nature. 1996;381:751–758. - PubMed
-
- Kelsoe G. Life and death in germinal centers (Redux) Immunity. 1996;4:107–111. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

