Characterization of an adenovirus vector containing a heterologous peptide epitope in the HI loop of the fiber knob
- PMID: 9499035
- PMCID: PMC109474
- DOI: 10.1128/JVI.72.3.1844-1852.1998
Characterization of an adenovirus vector containing a heterologous peptide epitope in the HI loop of the fiber knob
Abstract
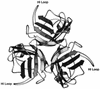
The utility of the present generation of recombinant adenovirus vectors for gene therapy applications could potentially be improved by designing targeted vectors capable of gene delivery to selected cell types in vivo. In order to achieve such targeting, we are investigating the possibilities of incorporation of ligands in the adenovirus fiber protein, which mediates primary binding of adenovirus to its cell surface receptor. Based on the proposed structure of the cell-binding domain of the fiber, we hypothesized that the HI loop of the fiber knob can be utilized as a convenient locale for incorporation of heterologous ligands. In this study, we utilized recombinant fiber proteins expressed in baculovirus-infected insect cells to demonstrate that the incorporation of the FLAG octapeptide into the HI loop does not ablate fiber trimerization and does not disturb formation of the cell-binding site localized in the knob. We then generated a recombinant adenovirus containing this modified fiber and showed that the short peptide sequence engineered in the knob is compatible with the biological functions of the fiber. In addition, by using a ligand-specific antibody, we have shown that the peptide incorporated into the knob remains available for binding in the context of mature virions containing modified fibers. These findings suggest that heterologous ligands can be incorporated into the HI loop of the fiber knob and that this locale possesses properties consistent with its employment in adenovirus retargeting strategies.
Figures

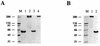




Similar articles
-
Engineering of adenovirus vectors containing heterologous peptide sequences in the C terminus of capsid protein IX.J Virol. 2002 Jul;76(14):6893-9. doi: 10.1128/jvi.76.14.6893-6899.2002. J Virol. 2002. PMID: 12072490 Free PMC article.
-
Tropism modification of adenovirus vectors by peptide ligand insertion into various positions of the adenovirus serotype 41 short-fiber knob domain.J Virol. 2007 Mar;81(6):2688-99. doi: 10.1128/JVI.02722-06. Epub 2006 Dec 27. J Virol. 2007. PMID: 17192304 Free PMC article.
-
Generation of fiber-modified adenovirus vectors containing heterologous peptides in both the HI loop and C terminus of the fiber knob.J Gene Med. 2003 Apr;5(4):267-76. doi: 10.1002/jgm.348. J Gene Med. 2003. PMID: 12692861
-
Tropism and transduction of oncolytic adenovirus 5 vectors in cancer therapy: Focus on fiber chimerism and mosaicism, hexon and pIX.Virus Res. 2018 Sep 15;257:40-51. doi: 10.1016/j.virusres.2018.08.012. Epub 2018 Aug 17. Virus Res. 2018. PMID: 30125593 Review.
-
Adenovirus interaction with its cellular receptor CAR.Curr Top Microbiol Immunol. 2003;272:331-64. doi: 10.1007/978-3-662-05597-7_11. Curr Top Microbiol Immunol. 2003. PMID: 12747555 Review.
Cited by
-
Adenovirus fiber shaft contains a trimerization element that supports peptide fusion for targeted gene delivery.J Virol. 2006 Dec;80(24):12324-31. doi: 10.1128/JVI.01331-06. Epub 2006 Oct 4. J Virol. 2006. PMID: 17020947 Free PMC article.
-
Antibody-mediated targeting of an adenovirus vector modified to contain a synthetic immunoglobulin g-binding domain in the capsid.J Virol. 2003 Feb;77(3):2093-104. doi: 10.1128/jvi.77.3.2093-2104.2003. J Virol. 2003. PMID: 12525644 Free PMC article.
-
Interferon-α Silencing by Small Interference RNA Increases Adenovirus Transduction and Transgene Expression in Huh7 Cells.Mol Biotechnol. 2018 Apr;60(4):251-258. doi: 10.1007/s12033-018-0066-7. Mol Biotechnol. 2018. PMID: 29478171
-
Genetic and Chemical Capsid Modifications of Adenovirus Vectors to Modulate Vector-Host Interactions.Viruses. 2021 Jul 2;13(7):1300. doi: 10.3390/v13071300. Viruses. 2021. PMID: 34372506 Free PMC article. Review.
-
In vivo targeted gene delivery using Adenovirus-antibody molecular glue conjugates.bioRxiv [Preprint]. 2025 Feb 1:2025.01.31.635969. doi: 10.1101/2025.01.31.635969. bioRxiv. 2025. Update in: Mol Ther Methods Clin Dev. 2025 May 26;33(2):101497. doi: 10.1016/j.omtm.2025.101497. PMID: 39974927 Free PMC article. Updated. Preprint.
References
-
- Belin M T, Boulanger P. Involvement of cellular adhesion sequences in the attachment of adenovirus to the HeLa cell surface. J Gen Virol. 1993;74:1485–1497. - PubMed
-
- Bergelson J M, Cunningham J A, Droguett G, Kurt-Jones E A, Krithivas A, Hong J S, Horwitz M S, Crowell R L, Finberg R W. Isolation of a common receptor for coxsackie B viruses and adenoviruses 2 and 5. Science. 1997;275:1320–1323. - PubMed
-
- Bout A, Imler J L, Schultz H, Perricaudet M, Zurcher C, Herbrink P, Valerio D, Pavirani A. In vivo adenovirus-mediated transfer of human CFTR cDNA to rhesus monkey airway epithelium: efficacy, toxicity and safety. Gene Ther. 1994;1:385–394. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

