The glycoprotein D (US6) homolog is not essential for oncogenicity or horizontal transmission of Marek's disease virus
- PMID: 9499123
- PMCID: PMC109562
- DOI: 10.1128/JVI.72.3.2548-2553.1998
The glycoprotein D (US6) homolog is not essential for oncogenicity or horizontal transmission of Marek's disease virus
Abstract
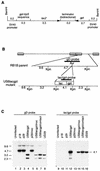
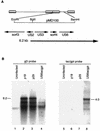
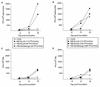
RB1BUS6lacgpt, a Marek's disease virus (MDV) mutant having a disrupted glycoprotein D (gD) homolog gene, established infection and induced tumors in chickens exposed to it by inoculation or by contact. Lymphoblastoid cell lines derived from RB1BUS6lacgpt-induced tumors harbored only the mutant virus. These results provide strong evidence that an intact gD homolog gene is not essential for oncogenicity or horizontal transmission of MDV.
Figures

Similar articles
-
Characterization of a Marek's disease virus mutant containing a lacZ insertion in the US6 (gD) homologue gene.Virus Genes. 1994 Sep;9(1):5-13. doi: 10.1007/BF01703430. Virus Genes. 1994. PMID: 7871761
-
Horizontal transmission of Marek's disease virus requires US2, the UL13 protein kinase, and gC.J Virol. 2007 Oct;81(19):10575-87. doi: 10.1128/JVI.01065-07. Epub 2007 Jul 18. J Virol. 2007. PMID: 17634222 Free PMC article.
-
The Tegument Protein pUL47 of Marek's Disease Virus Is Necessary for Horizontal Transmission and Is Important for Expression of Glycoprotein gC.J Virol. 2020 Dec 22;95(2):e01645-20. doi: 10.1128/JVI.01645-20. Print 2020 Dec 22. J Virol. 2020. PMID: 32999032 Free PMC article.
-
Latency and tumorigenesis in Marek's disease.Avian Dis. 2013 Jun;57(2 Suppl):360-5. doi: 10.1637/10470-121712-Reg.1. Avian Dis. 2013. PMID: 23901747 Review.
-
T-cell transformation by Marek's disease virus.Trends Microbiol. 1999 Jan;7(1):22-9. doi: 10.1016/s0966-842x(98)01427-9. Trends Microbiol. 1999. PMID: 10068994 Review.
Cited by
-
Transcriptional analysis of Marek's disease virus glycoprotein D, I, and E genes: gD expression is undetectable in cell culture.J Virol. 2001 Mar;75(5):2067-75. doi: 10.1128/JVI.75.5.2067-2075.2001. J Virol. 2001. PMID: 11160711 Free PMC article.
-
Marek's disease virus-1 unique gene LORF1 is involved in viral replication and MDV-1/Md5-induced atrophy of the bursa of Fabricius.PLoS Pathog. 2025 Feb 3;21(2):e1012891. doi: 10.1371/journal.ppat.1012891. eCollection 2025 Feb. PLoS Pathog. 2025. PMID: 39899476 Free PMC article.
-
Clustering of mutations within the inverted repeat regions of a serially passaged attenuated gallid herpesvirus type 2 strain.Virus Genes. 2008 Aug;37(1):69-80. doi: 10.1007/s11262-008-0242-0. Epub 2008 May 31. Virus Genes. 2008. PMID: 18516669
-
Identification of non-essential loci within the Meleagrid herpesvirus 1 genome.Virol J. 2015 Aug 27;12:130. doi: 10.1186/s12985-015-0362-9. Virol J. 2015. PMID: 26307059 Free PMC article.
-
Latest Insights into Unique Open Reading Frames Encoded by Unique Long (UL) and Short (US) Regions of Marek's Disease Virus.Viruses. 2021 May 25;13(6):974. doi: 10.3390/v13060974. Viruses. 2021. PMID: 34070255 Free PMC article. Review.
References
-
- Audonnet J-C, Winslow J, Allen G, Paoletti E. Equine herpesvirus type 1 unique short fragment encodes glycoproteins with homology to herpes simplex virus type 1 gD, gI and gE. J Gen Virol. 1991;71:2969–2978. - PubMed
-
- Buckmaster A E, Scott S D, Sanderson M J, Boursnell M E G, Ross N L J, Binns M M. Gene sequence and mapping data from Marek’s disease virus and herpesvirus of turkeys: implication for herpesvirus classification. J Gen Virol. 1988;69:2033–2042. - PubMed
-
- Calnek B W, Witter R L. Marek’s disease. In: Calnek B W, editor. Diseases of poultry. Ames: Iowa State University Press; 1997. pp. 369–413.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

