Recovery from desensitization of neuronal nicotinic acetylcholine receptors of rat chromaffin cells is modulated by intracellular calcium through distinct second messengers
- PMID: 9502806
- PMCID: PMC6793119
- DOI: 10.1523/JNEUROSCI.18-07-02458.1998
Recovery from desensitization of neuronal nicotinic acetylcholine receptors of rat chromaffin cells is modulated by intracellular calcium through distinct second messengers
Abstract
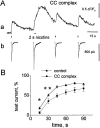
The mechanisms through which changes in intracellular Ca2+ concentration ([Ca2+]i) might influence desensitization of neuronal nicotinic receptors (nAChRs) of rat chromaffin cells were investigated by simultaneous patch-clamp recording of membrane currents and confocal microscopy imaging of [Ca2+]i induced by nicotine. Increases in [Ca2+]i that were induced by membrane depolarization or occurred spontaneously did not influence inward currents elicited by focally applied test pulses (10 msec) of nicotine, indicating that raised [Ca2+]i per se did not trigger desensitization of nAChRs. Desensitization of nAChRs, evoked by 2 sec focal application of nicotine, which largely raised [Ca2+]i, was not affected by intracellular application of agents that activate or depress protein kinase C (PKC) or A (PKA) or inhibit phosphatase 1, 2 A and B. Conversely, recovery from desensitization was facilitated by the phorbol ester phorbol 12-myristate 13-acetate (PMA) or the phosphatase 2 B inhibiting complex of cyclosporin A-cyclophilin A, whereas it was impaired by the broad spectrum kinase inhibitor staurosporine. The effects of PMA or staurosporine were prevented by the intracellularly applied Ca2+ chelator BAPTA. The adenylate cyclase activator forskolin accelerated recovery, whereas the selective PKA antagonist Rp-cAMPS had an opposite effect. The action of staurosporine and Rp-cAMPS on recovery from desensitization was additive. It is proposed that when nAChRs are desensitized, they become susceptible to modulation by [Ca2+]i via intracellular second messengers such as serine/threonine kinases and calcineurin. Thus, the phosphorylation state of neuronal nAChRs appears to regulate their rate of recovery from desensitization.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
