Distinct ontogeny of glucocorticoid and mineralocorticoid receptor and 11beta-hydroxysteroid dehydrogenase types I and II mRNAs in the fetal rat brain suggest a complex control of glucocorticoid actions
- PMID: 9502816
- PMCID: PMC6793118
- DOI: 10.1523/JNEUROSCI.18-07-02570.1998
Distinct ontogeny of glucocorticoid and mineralocorticoid receptor and 11beta-hydroxysteroid dehydrogenase types I and II mRNAs in the fetal rat brain suggest a complex control of glucocorticoid actions
Abstract
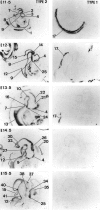
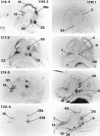
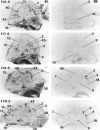
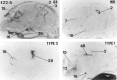
Glucocorticoids (GCs) act via intracellular mineralocorticoid (MR) and glucocorticoid receptors (GR). However, it has recently been recognized that GC access to receptors is determined by the presence of tissue-specific 11beta-hydroxysteroid dehydrogenases (11beta-HSDs) that catalyze the interconversion of active corticosterone and inert 11-dehydrocorticosterone. 11beta-HSD type 1 (11beta-HSD1) is a bidirectional enzyme in vitro that acts predominantly as a reductase (regenerating corticosterone) in intact neurons. In contrast, 11beta-HSD type 2 (11beta-HSD2) is a higher affinity exclusive dehydrogenase that excludes GCs from MR in the kidney, producing aldosterone-selectivity in vivo. We have examined the ontogeny of 11beta-HSD mRNAs and enzyme activity during prenatal brain development and correlated this with GR and MR mRNA development. These data reveal that (1) 11beta-HSD2 mRNA is highly expressed in all CNS regions during midgestation, but expression is dramatically reduced during the third trimester except in the thalamus and cerebellum; (2) 11beta-HSD2-like activity parallels closely the pattern of mRNA expression; (3) 11beta-HSD1 mRNA is absent from the CNS until the the third trimester, and activity is low or undectectable; and (4) GR mRNA is highly expressed throughout the brain from midgestation, but MR gene expression is absent until the last few days of gestation. High 11beta-HSD2 at midgestation may protect the developing brain from activation of GR by GCs. Late in gestation, repression of 11beta-HSD2 gene expression may allow increasing GC activation of GR and MR, permitting key GC-dependent neuronal and glial maturational events.
Figures



References
-
- Ader R, Blackman DE. Prenatal psychological stress and offspring behavior in rats and mice. Science. 1971;125:698–699. - PubMed
-
- Agarwal A, Monder C, Eckstein B, White PC. Cloning and expression of rat cDNA encoding corticosteroid 11-dehydrogenase. J Biol Chem. 1989;264:18939–18943. - PubMed
-
- Altman J, Bayer SA. Atlas of prenatal rat brain development. CRC; London: 1995.
-
- Arriza JL, Simerly RB, Swanson LW, Evans RM. The neuronal mineralocorticoid receptor as a mediator of glucocorticoid response. Neuron. 1988;1:887–900. - PubMed
-
- Barlow SM, Knight AF, Sullivan FM. Delay in postnatal growth and development of offspring produced by maternal restraint stress during pregnancy in the rat. Teratology. 1978;18:211–218. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
