Ubiquitin is covalently attached to the p6Gag proteins of human immunodeficiency virus type 1 and simian immunodeficiency virus and to the p12Gag protein of Moloney murine leukemia virus
- PMID: 9525617
- PMCID: PMC109742
- DOI: 10.1128/JVI.72.4.2962-2968.1998
Ubiquitin is covalently attached to the p6Gag proteins of human immunodeficiency virus type 1 and simian immunodeficiency virus and to the p12Gag protein of Moloney murine leukemia virus
Abstract
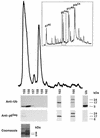
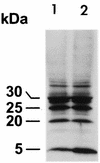
Host proteins are incorporated into retroviral virions during assembly and budding. We have examined three retroviruses, human immunodeficiency virus type 1 (HIV-1), simian immunodeficiency virus (SIV), and Moloney murine leukemia virus (Mo-MuLV), for the presence of ubiquitin inside each of these virions. After a protease treatment to remove exterior viral as well as contaminating cellular proteins, the proteins remaining inside the virion were analyzed. The results presented here show that all three virions incorporate ubiquitin molecules at approximately 10% of the level of Gag found in virions. In addition to free ubiquitin, covalent ubiquitin-Gag complexes were detected, isolated, and characterized from all three viruses. Our immunoblot and protein sequencing results on treated virions showed that approximately 2% of either HIV-1 or SIV p6Gag was covalently attached to a single ubiquitin molecule inside the respective virions and that approximately 2 to 5% of the p12Gag in Mo-MuLV virions was monoubiquitinated. These results show that ubiquitination of Gag is conserved among these retroviruses and occurs in the p6Gag portion of the Gag polyprotein, a region that is likely to be involved in assembly and budding.
Figures
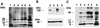
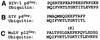

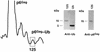
References
-
- Arthur L O, Bess J W, Jr, Sowder II R C, Benveniste R E, Mann L D, Chermann J-C, Henderson L E. Cellular proteins bound to immunodeficiency viruses: implications for pathogenesis and vaccines. Science. 1992;258:1935–1938. - PubMed
-
- Ball E, Karlik C C, Beall C J, Saville D L, Sparrow J C, Bullard B, Fyrberg E A. Arthrin, a myofibrillar protein of insect flight muscle, is an actin-ubiquitin conjugate. Cell. 1987;51:221–228. - PubMed
-
- Bess J W, Jr, Gorelick R J, Bosche W J, Henderson L E, Arthur L O. Microvesicles are a source of contaminating cellular proteins found in purified HIV-1 preparations. Virology. 1997;230:134–144. - PubMed
-
- Chau V, Tobias J W, Bachmair A, Marriott D, Ecker D J, Gonda D K, Varshavsky A. A multiubiquitin chain is confined to specific lysine in a targeted short-lived protein. Nature. 1989;243:1576–1583. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

