Inhibitory effect of 2'-fluoro-5-methyl-beta-L-arabinofuranosyl-uracil on duck hepatitis B virus replication
- PMID: 9527788
- PMCID: PMC105416
- DOI: 10.1128/AAC.42.2.369
Inhibitory effect of 2'-fluoro-5-methyl-beta-L-arabinofuranosyl-uracil on duck hepatitis B virus replication
Abstract
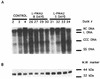
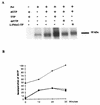
The antiviral activity of 2'-fluoro-5-methyl-beta-L-arabinofuranosyluracil (L-FMAU), a novel L-nucleoside analog of thymidine known to be an inhibitor of hepatitis B virus (HBV) replication in hepatoma cells (2.2.1.5 cell line), was evaluated in the duck HBV (DHBV) model. Short-term oral administration (5 days) of L-FMAU (40 mg/kg of body weight/day) to experimentally infected ducklings induced a significant decrease in the level of viremia. This antiviral effect was sustained in animals when therapy was prolonged for 8 days. The histological study showed no evidence of liver toxicity in the L-FMAU-treated group. By contrast, microvesicular steatosis was found in the livers of dideoxycytidine-treated animals. L-FMAU administration in primary duck hepatocyte cultures infected with DHBV induced a dose-dependent inhibition of both virion release in culture supernatants and intracellular viral DNA synthesis, without clearance of viral covalently closed circular DNA. By using a cell-free system for the expression of an enzymatically active DHBV reverse transcriptase, it was shown that L-FMAU triphosphate exhibits an inhibitory effect on the incorporation of dAMP in the viral DNA primer. Thus, our data demonstrate that L-FMAU inhibits DHBV replication in vitro and in vivo. Long-term administration of L-FMAU for the eradication of viral infection in animal models of HBV infection should be evaluated.
Figures




References
-
- Bartholomew M, Jansen R, Jeffers L, Reddy K, Johnson L, Bunzendahl H, Condreay L, Tzakis A, Schiff A, Brown N. Hepatitis B virus resistance to lamivudine given for recurrent infection after orthotopic liver transplantation. Lancet. 1997;349:20–22. - PubMed
-
- Bridges E, Cheng Y C. Use of novel β-l(−)-nucleoside analogues for treatment and prevention of chronic hepatitis B virus infection and hepatocellular carcinoma. Prog Liver Dis. 1995;13:231–245. - PubMed
-
- Chassot S, Lambert V, Kay A, Godinot C, Trépo C, Cova L. Identification of major antigenic domains of duck hepatitis B virus pre-S protein by peptide scanning. Virology. 1994;200:72–78. - PubMed
-
- Chen C, Cheng Y C. Delayed cytoxicity and selective loss of mitochondrial DNA in cells treated with the anti-human immunodeficiency virus compound 2′,3′-dideoxycytidine. J Biol Chem. 1989;264:11934–11937. - PubMed
-
- Cheng, Y.-C. Unpublished data.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

